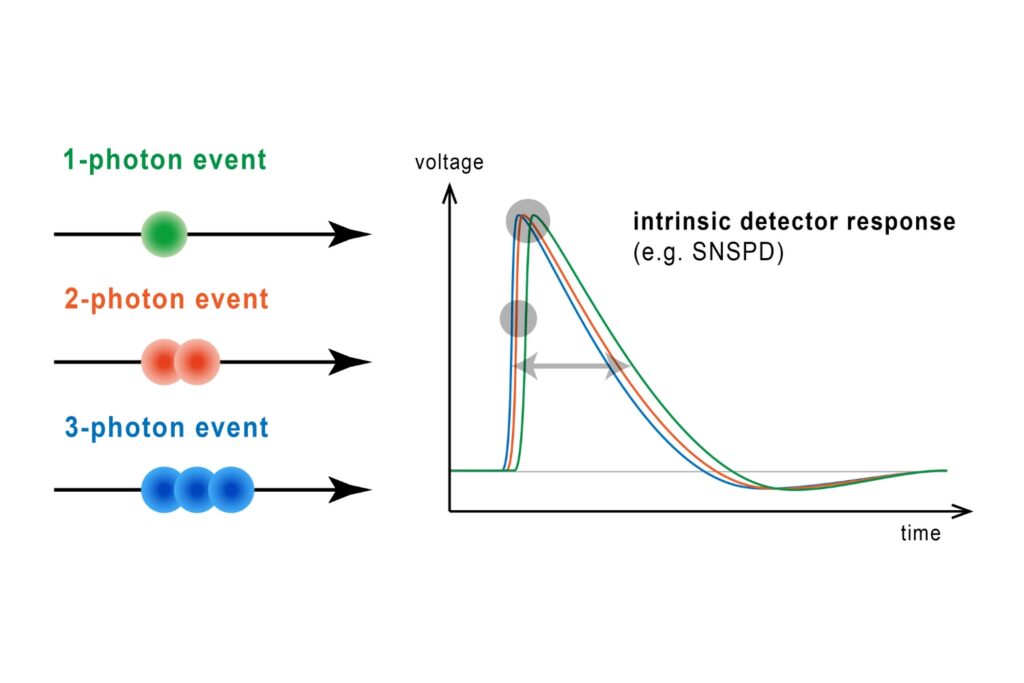
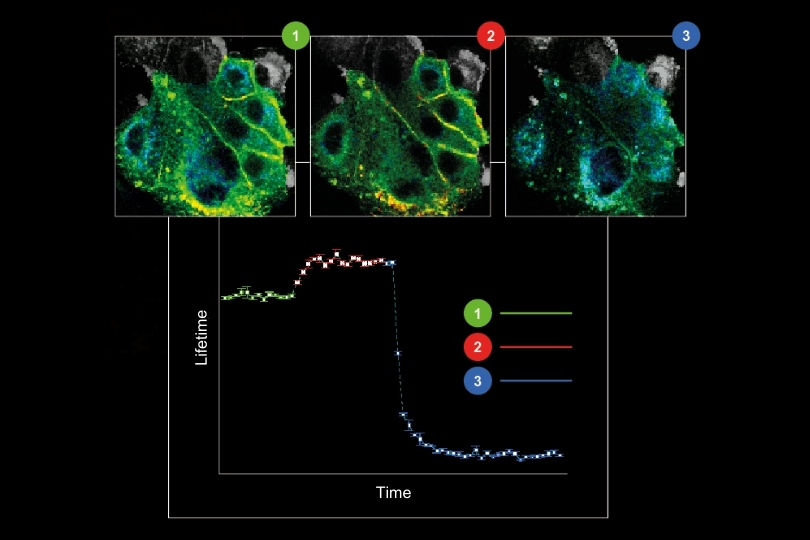
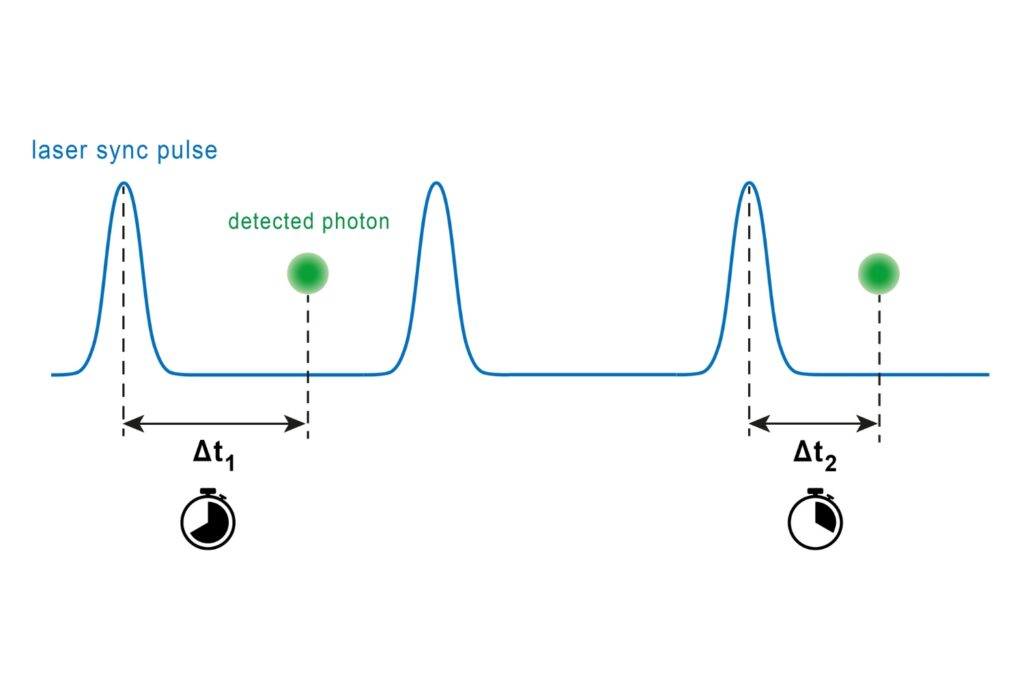
The HydraHarp 400 Multichannel Picosecond Event Timer & TCSPC Module has been discontinued as of November 2025. This product is no longer available for new orders.
Users looking for a comparable and updated solution are encouraged to consider its successor, the HydraHarp 500 high-resolution multichannel time tagger & TCSPC unit. The HydraHarp 500 provides advanced time-tagging and TCSPC capabilities and is the recommended replacement for applications previously implemented with the HydraHarp 400.
Explore the HydraHarp 500 product page for detailed specifications and features.