Photon Counting Detectors


Simplify your materials characterization with one flexible TRPL microscope enabling multiple methods for precise and efficient analysis.

Complete confocal fluorescence microscope that empowers researchers to advance quantitative functional imaging from individual molecules to cells and tissues.

Compact FLIM and FCS upgrade kit that adds advanced functional imaging and correlation analysis to existing laser scanning microscopes.

Designed for flexible, sensitive, and precise steady-state and time-resolved spectroscopy across the UV to NIR range and time scales from picoseconds to milliseconds.

Modular lifetime spectrometer designed for flexible fluorescence and photoluminescence measurements in both materials and life science research.

Add spectral and time-resolved photoluminescence to your setup through flexible microscope–spectrometer coupling options.

Get the most out of superconducting nanowire detectors in large-scale quantum communication and computing experiments requiring precise multichannel timing.

Boost your time-resolved experiments with a flexible, high-precision time tagging and TCSPC unit for materials science and quantum sensing.

Scale your photonic quantum computing and detector characterization setups while maintaining performance, flexibility, and high data throughput.

Compact 3-color picosecond laser delivering flexible ns to ms excitation with cost-effective multicolor performance and straightforward operation.

Smart picosecond laser diode heads covering UV-A to NIR, providing the right combination of power, pulse width, and diode type for any time-resolved technique.

VisUV provides clean short pulses and stable timing across key UV and visible wavelengths, including deep UV lines as well as 488 nm and 532 nm.

Enhance your single-photon counting experiments with wide dynamic range and excellent timing precision in the UV and visible even at the highest count rates.

Capture even the weakest signals over large areas with maximum dynamic range and enhanced low-light sensitivity in a compact detector design.

Unlock spatially resolved single-photon detection with a 23-pixel SPAD array, combining low dark counts and precise time tagging for advanced experiments.
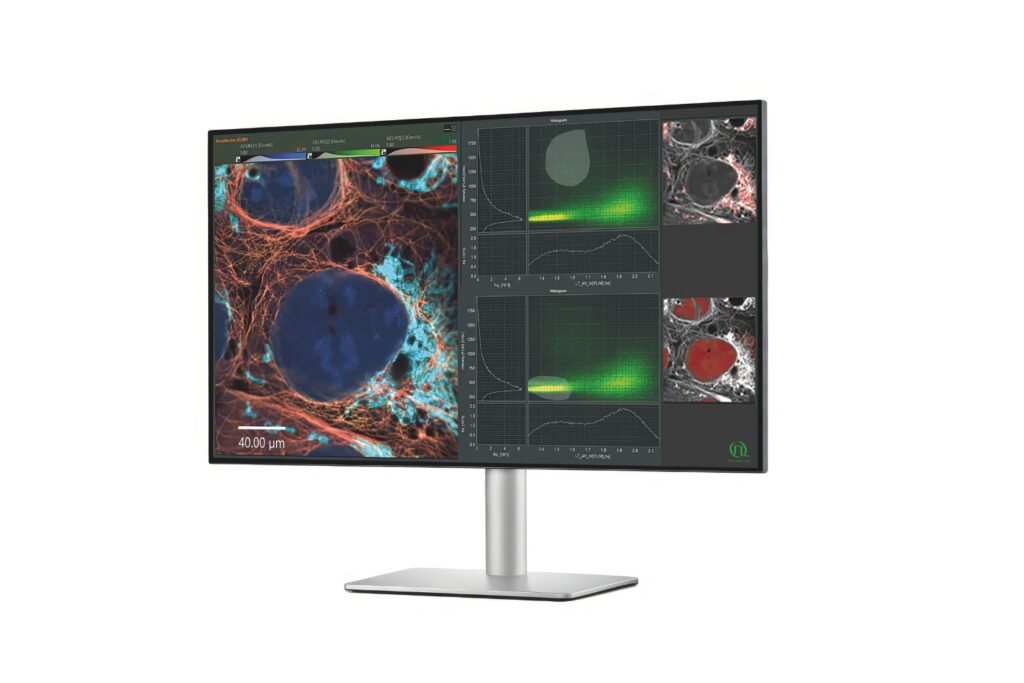
Advanced FLIM analysis software for fast, accurate interpretation of lifetime imaging data.
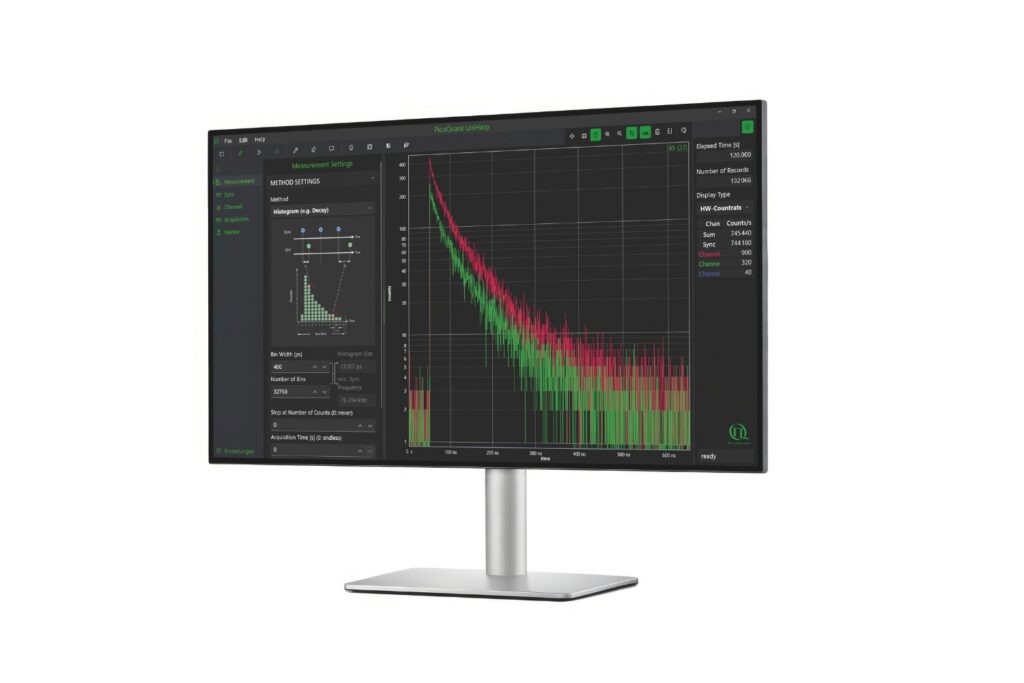
Intuitive, free software solution for real-time, high-precision photon data acquisition, visualization, and initial data analysis.
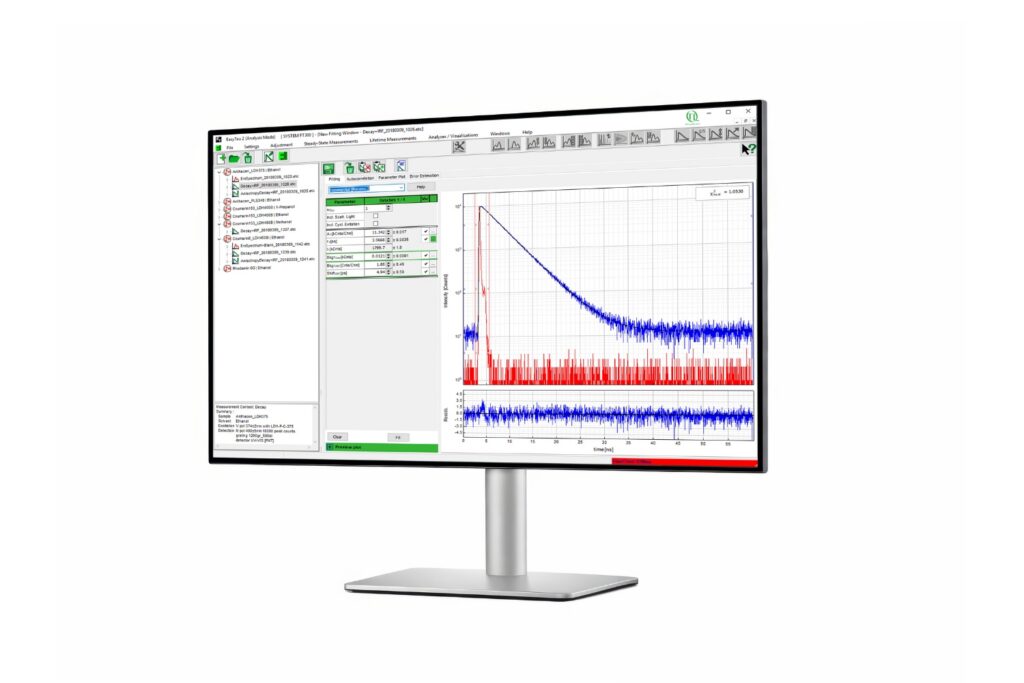
Advanced software for time-resolved fluorescence acquisition and analysis.

An imaging technique that uses fluorescence lifetimes to generate image contrast.
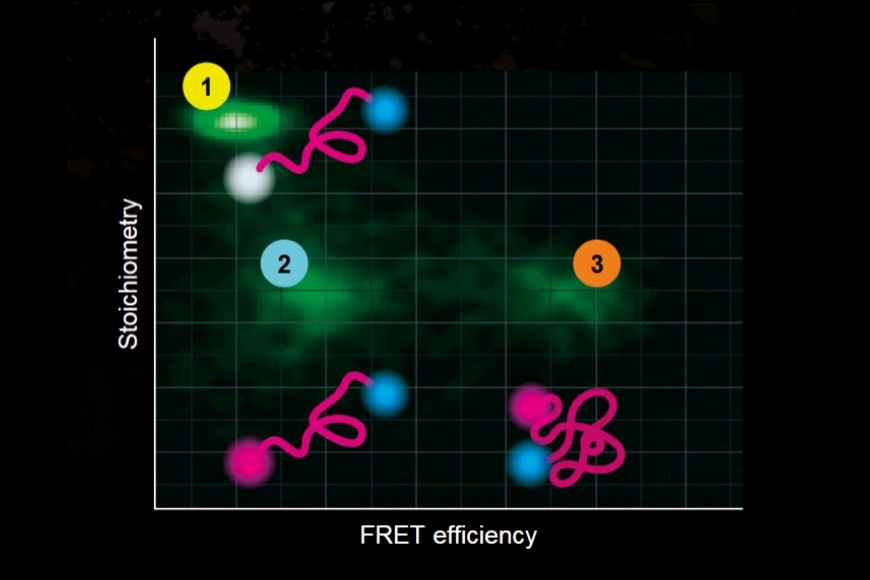
Investigating how proteins dynamically explore multiple conformational states that control biological function.

Investigating how biomolecules separate into dynamic liquid phases to organize cellular space and regulate biological function.
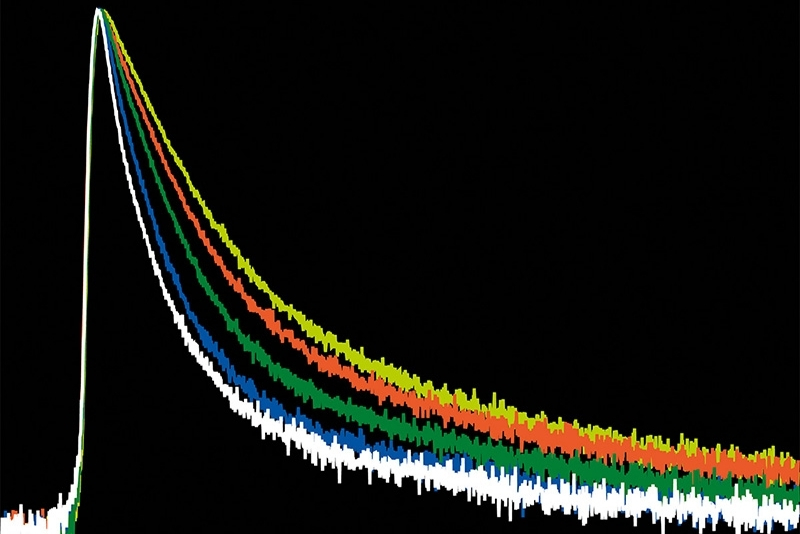
A time-resolved technique that measures photoluminescence lifetimes to reveal excited-state dynamics in materials.
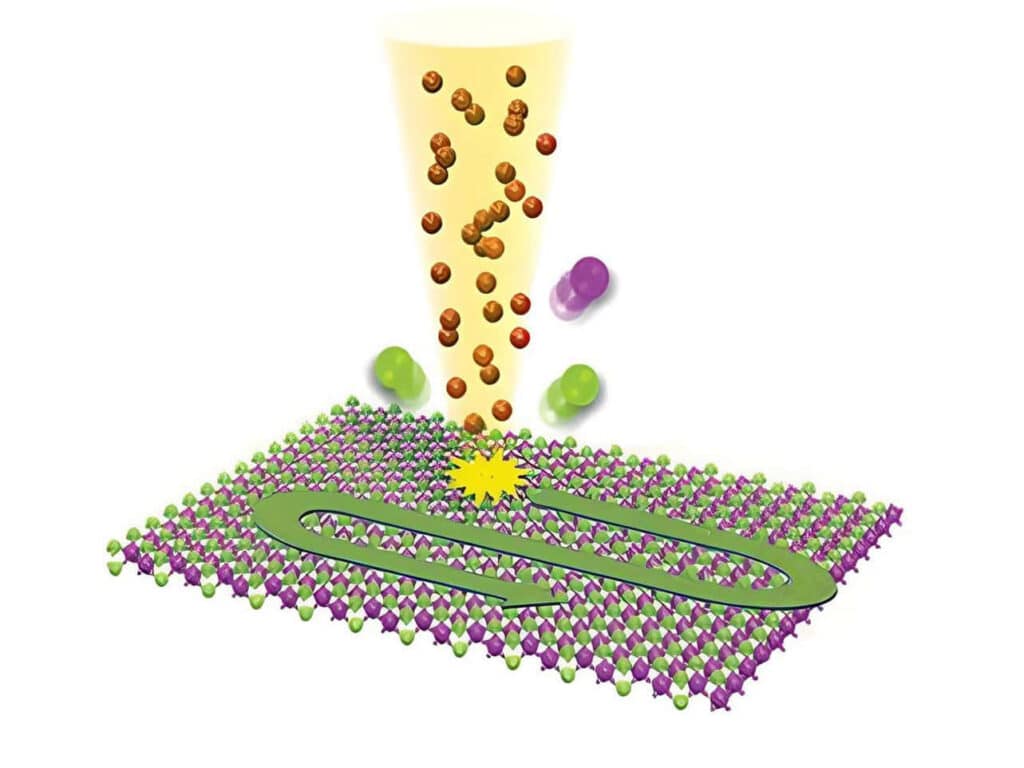
Studying exciton dynamics, charge carrier processes, and structural properties through optical and time-resolved characterization methods.

Investigating charge-carrier lifetimes and recombination dynamics to enable precise optical characterization of material quality and device performance.
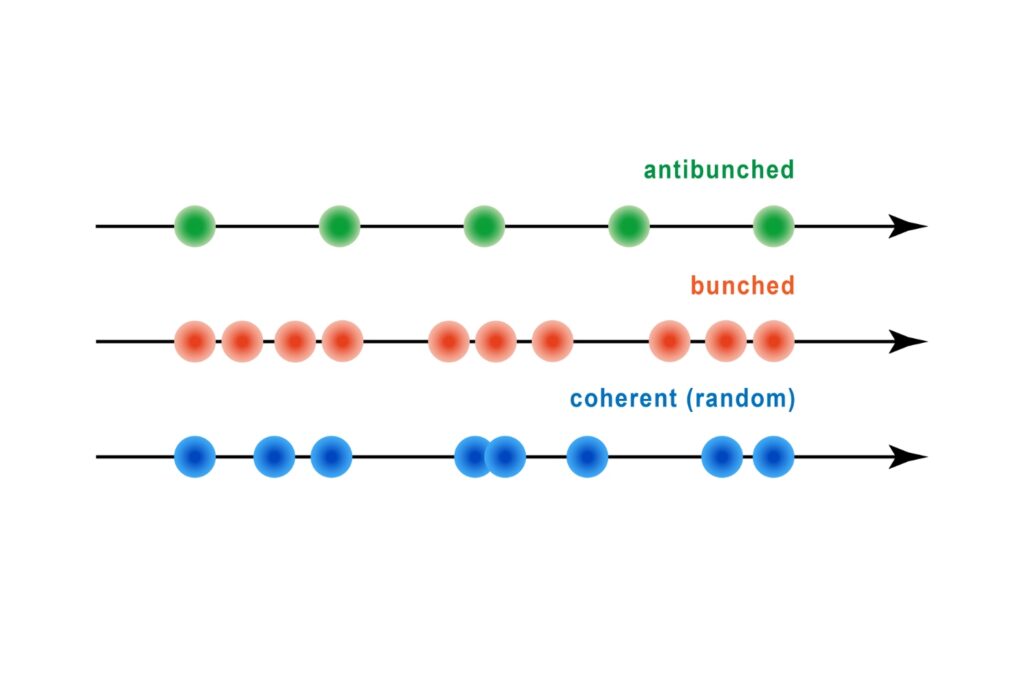
A quantum optical signature revealed by time-resolved photon correlation analysis to identify single-photon emission in materials and nanostructures.
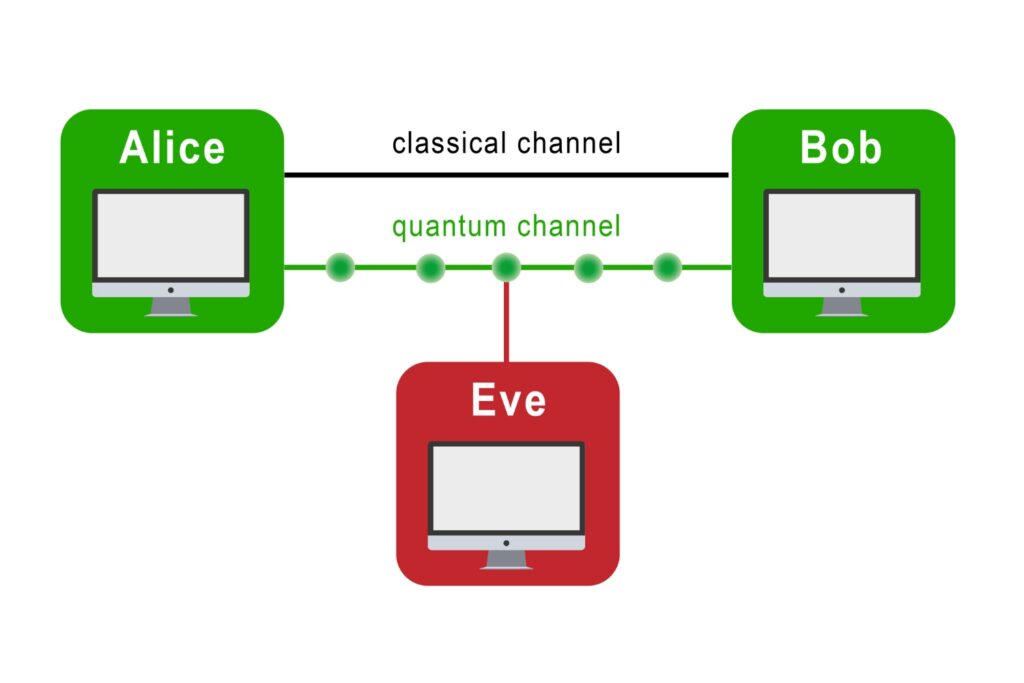
The transmission of information using individual photons, using quantum effects to ensure absolute security.
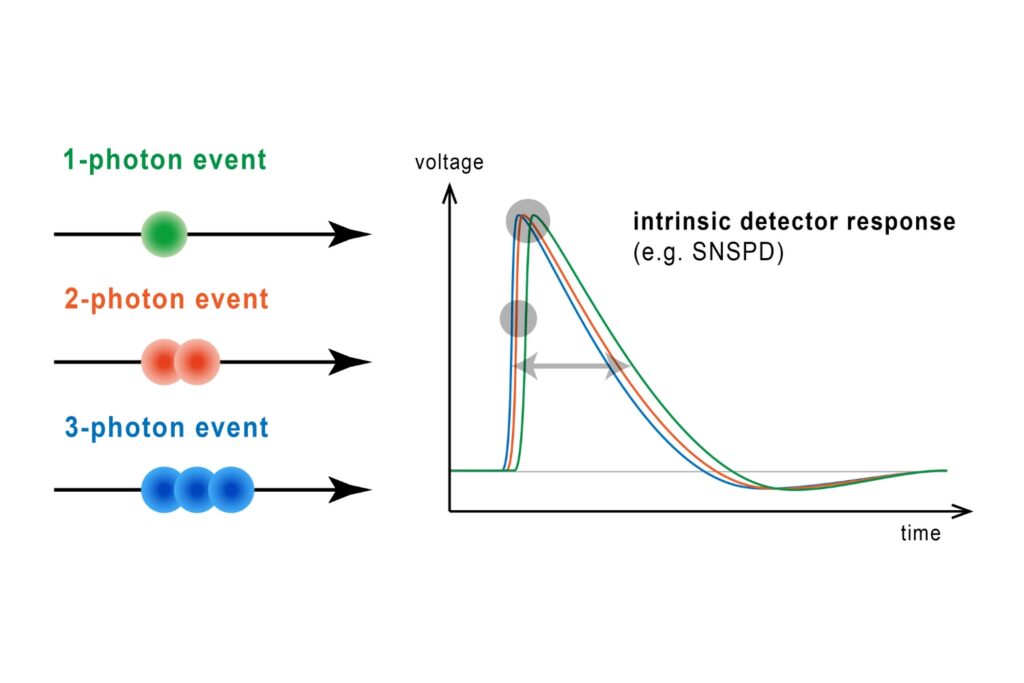
Quantifying photons per detection event enables direct access to photon-number statistics, providing insight into quantum and statistical properties of light.

An optical technique that analyzes light emission under electrical excitation to reveal electronic properties of electroluminescent materials.
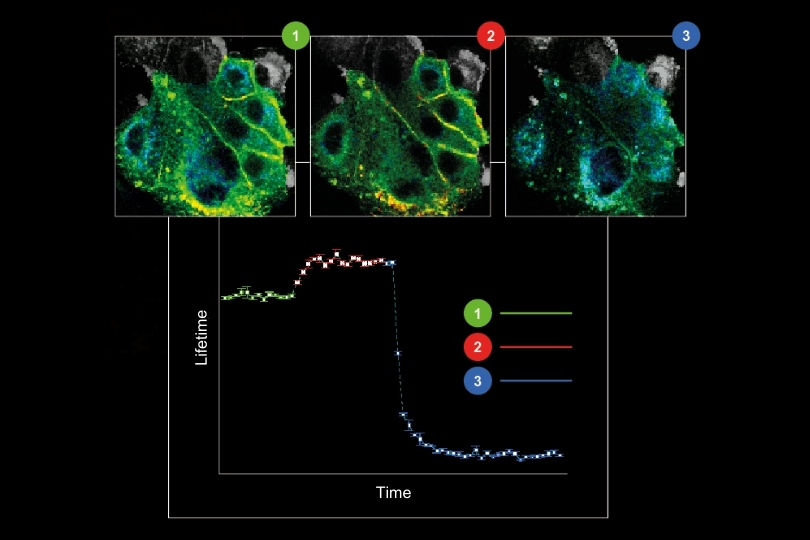
Monitoring environmental signals and trace compounds to understand dynamic changes in natural and engineered environments.
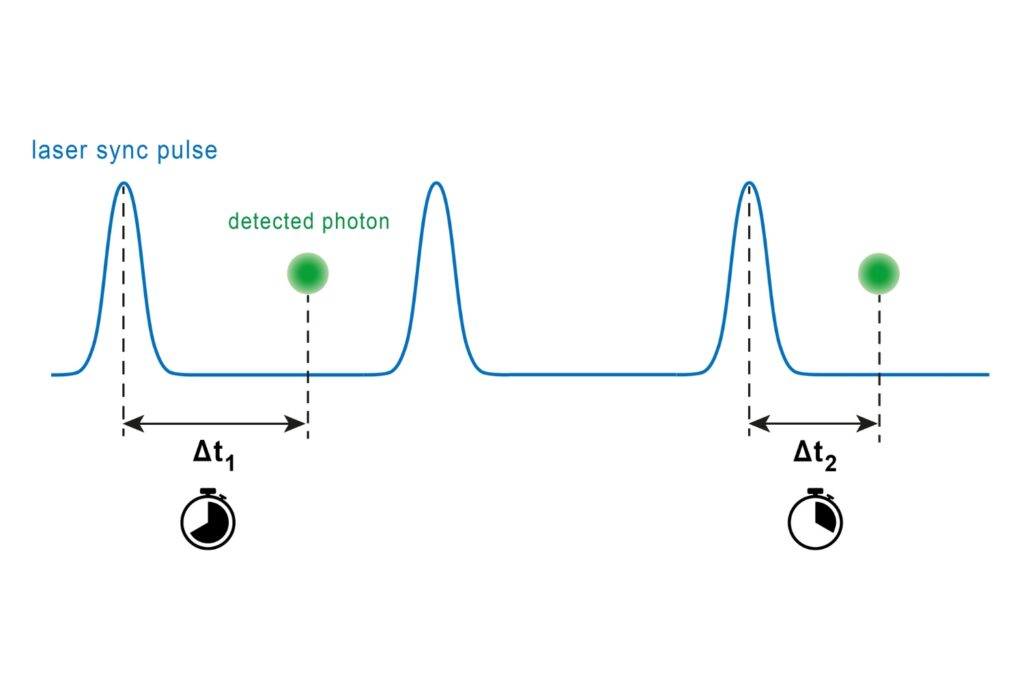
A photon timing technique that measures single-photon arrival times to resolve ultrafast dynamics in fluorescence, materials research, and quantum optics.
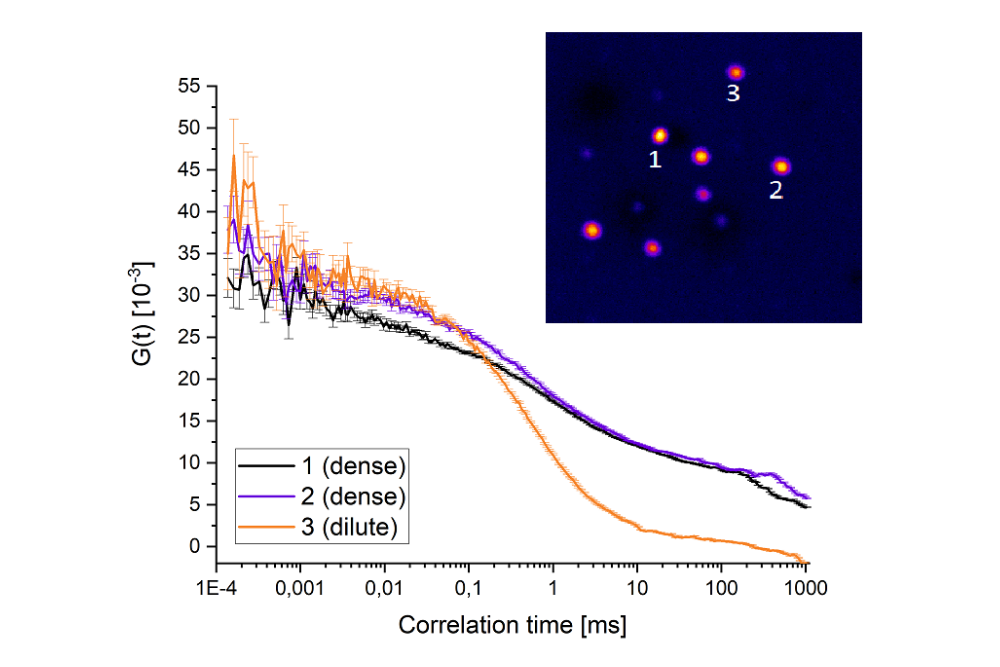
Fluorescence Correlation Spectroscopy (FCS) is a quantitative fluorescence spectroscopy technique that investigates molecular dynamics by analyzing temporal fluctuations in fluorescence intensity within a small, optically defined observation volume. These fluctuations arise as fluorescent molecules randomly diffuse into and out of the focal volume, causing time-dependent variations in the detected signal.
By statistically evaluating these fluorescence fluctuations, FCS provides access to dynamic information on molecular mobility, concentration and interactions. Because the method relies on correlation analysis rather than absolute signal intensity, FCS enables highly sensitive and quantitative measurements of molecular processes in solution, membranes, and living cells.
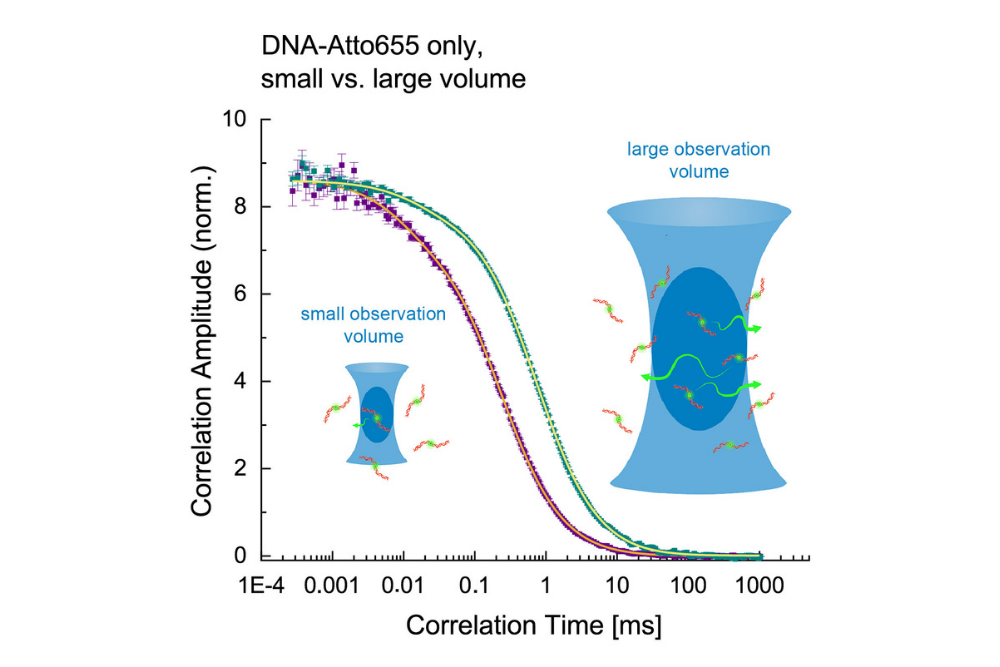 Comparison of small and large observation volumes in FCS. A larger volume shifts correlation curves to longer lag times, enabling measurement of slower diffusion processes.
Comparison of small and large observation volumes in FCS. A larger volume shifts correlation curves to longer lag times, enabling measurement of slower diffusion processes.Fluorescence Correlation Spectroscopy (FCS) measures spontaneous fluctuations in fluorescence intensity that occur as fluorescent molecules diffuse through a small, optically defined observation volume. These fluctuations are recorded as a time-dependent fluorescence signal and reflect molecular diffusion, reactions, or conformational changes.
The recorded intensity trace is evaluated using correlation analysis , which relates fluorescence fluctuations at different time points to extract information on molecular motion and interactions. Compared to steady-state or intensity-based approaches, FCS thus provides a sensitive, quantitative description of molecular dynamics based on time-dependent fluorescence fluctuations rather than absolute signal levels.
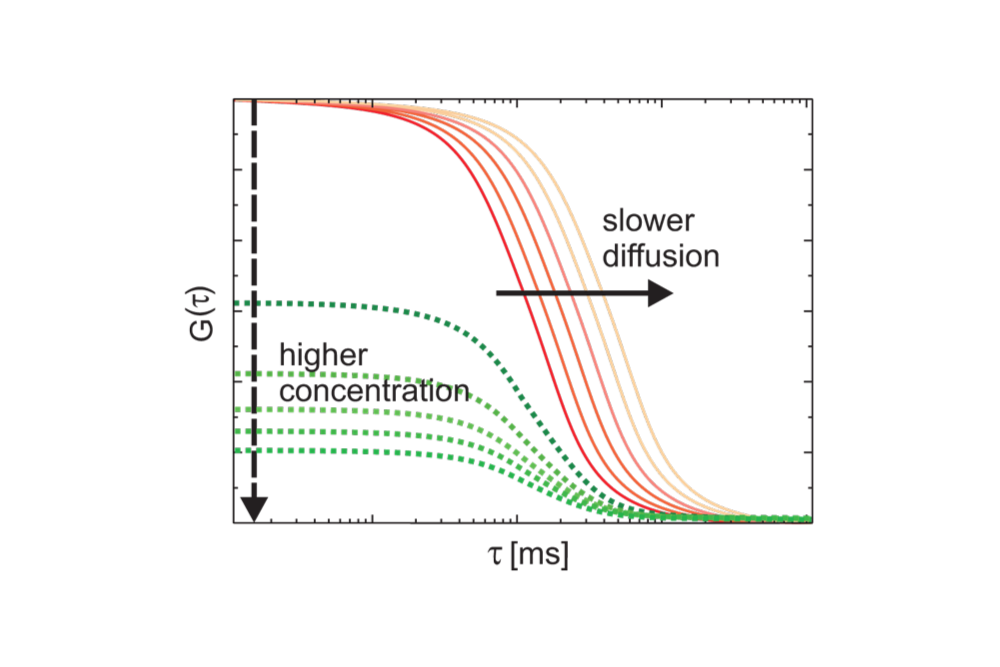 Illustration of how diffusion time and molecular concentration influence the autocorrelation function in Fluorescence Correlation Spectroscopy (FCS). Slower diffusion shifts the curve to longer lag times, while higher concentration reduces correlation amplitude.
Illustration of how diffusion time and molecular concentration influence the autocorrelation function in Fluorescence Correlation Spectroscopy (FCS). Slower diffusion shifts the curve to longer lag times, while higher concentration reduces correlation amplitude.Fluorescence Correlation Spectroscopy (FCS) enables quantitative analysis of molecular dynamics at very low concentrations, down to the single-molecule level. By analyzing fluorescence fluctuations rather than absolute signal intensity, FCS provides high sensitivity and robustness against intensity variations. The method allows direct access to diffusion behavior, concentrations, and molecular interactions in solution, membranes, and living cells. Because measurements are performed under equilibrium conditions and require minimal sample volumes, FCS is particularly well suited for studying fast, dynamic processes in complex biological systems.
The fluorescence intensity fluctuations recorded in FCS are evaluated using correlation functions to extract quantitative information on molecular dynamics. The central result of an FCS measurement is the temporal autocorrelation function, which describes how fluorescence fluctuations at one time point correlate with those at later times. The shape and amplitude of this correlation function reflect the underlying physical processes governing molecular motion and interactions within the observation volume.
Several complementary analysis approaches are commonly used in FCS, depending on experimental conditions and the desired information of interest:
These analysis strategies enable FCS to quantify molecular mobility, concentrations, and interactions over a wide range of temporal scales, depending on the detection method and the molecular system studied.
Dedicated software solutions such as the Luminosa software support FCS and FLCS analysis, offering correlation calculation, model-based fitting, and reproducible data evaluation within an integrated workflow.
 Luminosa single photon counting confocal fluorescence microscope designed for quantitative time-resolved and single-molecule imaging.
Luminosa single photon counting confocal fluorescence microscope designed for quantitative time-resolved and single-molecule imaging.Reliable Fluorescence Correlation Spectroscopy (FCS) measurements require precise optical control, sensitive photon detection, and stable time-resolved data acquisition. Because FCS analyzes fluorescence intensity fluctuations within a very small observation volume, instrument performance directly impacts the accuracy of correlation analysis.
Key instrumentation requirements include:
Scanning Fluorescence Correlation Spectroscopy (sFCS) extends FCS by scanning the observation volume along a defined path. This approach improves statistical robustness, reduces photobleaching, and enables reliable correlation analysis in heterogeneous or structured environments such as membranes and living cells.
Fluorescence Lifetime Correlation Spectroscopy (FLCS) combines FCS with fluorescence lifetime information to selectively correlate photons based on their decay characteristics. This allows efficient suppression of background signals, detector artifacts, or spectrally overlapping species, improving accuracy under challenging conditions.
Dual-focus Fluorescence Correlation Spectroscopy (2fFCS) uses two laterally shifted excitation volumes at a fixed distance to enable absolute, calibration-free diffusion measurements. Cross-correlating both excitation volumes reduces sensitivity to optical alignment and system-dependent parameters.
Fluorescence Cross-Correlation Spectroscopy (FCCS) extends FCS to multiple detection channels to study interactions between differently labeled molecules. Cross-correlation analysis reveals co-diffusion and molecular complex formation in solution and biological systems.
Fluorescence Correlation Spectroscopy (FCS) was used to measure the diffusion of GFP-Ago2 in ER293 cells. Using an LSM Upgrade Kit, FCS curves acquired in the nucleus and cytoplasm revealed distinct diffusion coefficients, indicating size differences between nuclear and cytosolic RISC complexes.
Fluorescence Correlation Spectroscopy (FCS) was performed at 266 nm using a MicroTime 200 with UV adaptation. High-quantum-efficiency laser dyes and streptavidin-coated beads were measured to demonstrate proof-of-principle FCS detection of intrinsic and UV-excited fluorescence.

Information bundle on fluorescence correlation spectroscopy (FCS) covering quantitative FCS, liFCS, dual-focus FCS, FLCS, calibration methods, and diffusion analysis.
In this customer video, Ben Schuler (University of Zürich) shows how single-molecule FRET combined with correlation spectroscopy and microfluidic mixing reveals the dynamics of intrinsically disordered proteins across timescales and environments.
Poster on next-generation TCSPC detection using PMA Hybrid detectors, enabling artifact-free FCS, antibunching measurements, and high-sensitivity FLIM imaging.
Please fill out the form below to receive the requested file. After submitting your details, the file will be sent to you by email.
* Required
Please fill out the form below to request more information. You may also use it to inquire about pricing, availability, technical specifications, or discuss your specific application. Our sales team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information about our products and services. You may also use it to ask for pricing, availability, technical specifications, or any other details relevant to your inquiry. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information and prices about our product. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required