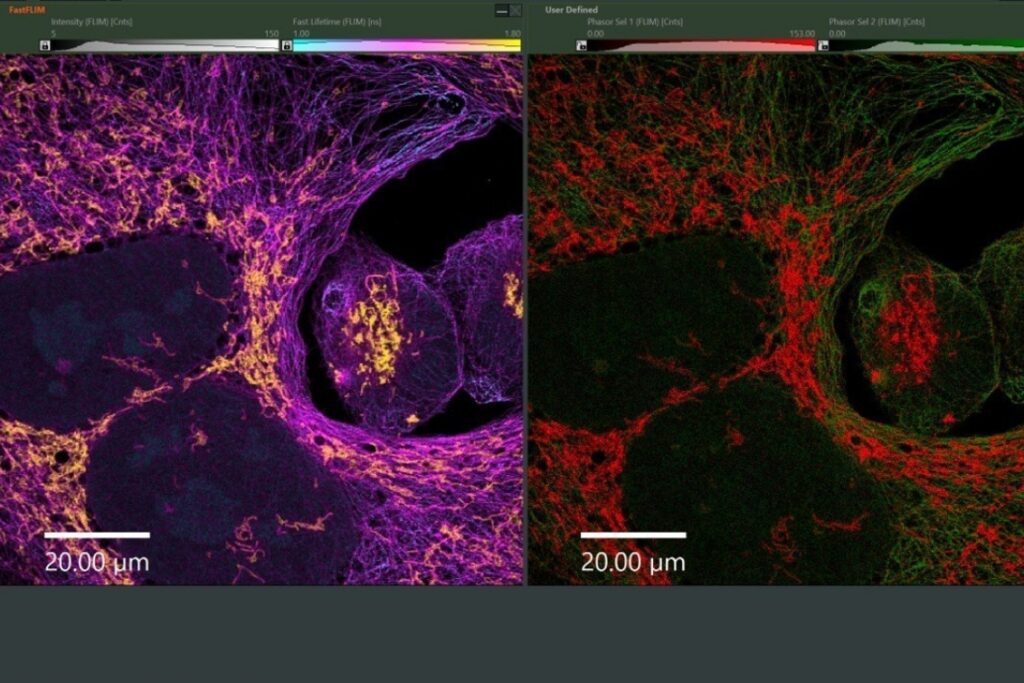
Modularity as Structural Design, Not as Extension
In many research environments, experimental setups evolve over time. New excitation schemes are introduced, additional detection channels become necessary, or external instruments need to be integrated.
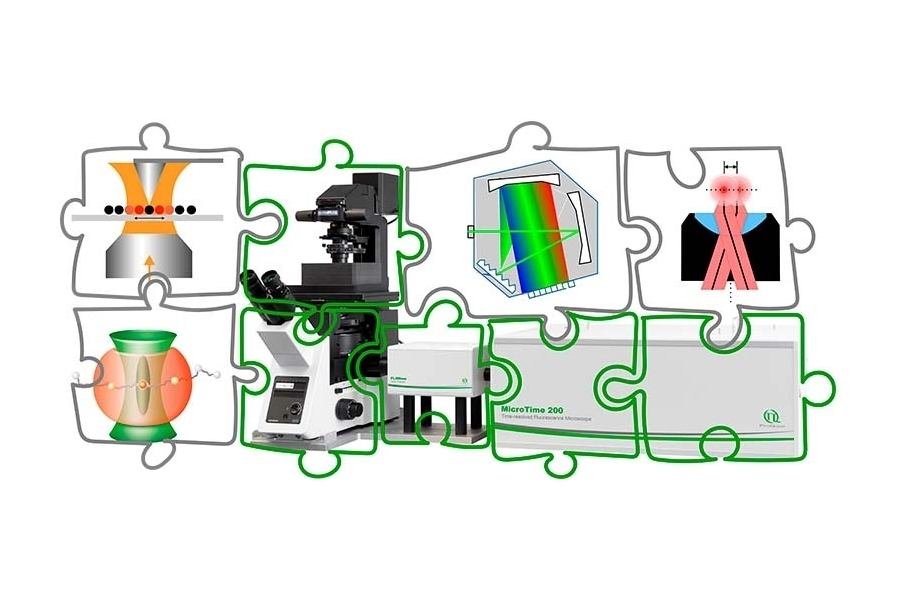
The MicroTime 200 is configured as a modular time-resolved confocal microscope with single molecule sensitivity in which excitation, detection, scanning, and timing electronics can be defined independently. This separation allows the system to be adapted to specific experimental constraints rather than restricting the experiment to a predefined configuration.
The following examples illustrate how different research groups have implemented customized system architectures based on their scientific requirements.
Correlated AFM and Fluorescence Lifetime Imaging in Functional Materials
Ignacio Martin-Fabiani – Loughborough University, UK
In functional materials research, optical properties often need to be correlated with structural and mechanical information on the nanoscale.
To achieve this, the MicroTime 200 (microscope body: Olympus IX73) was coupled with a Bruker BioScope Resolve atomic force microscope. The configuration included:
- Excitation at 405, 485, 530, and 640 nm
- Detectors: two Excelitas SPADs and two PMA Hybrid-40
- Time Tagging & TCSPC Electronics: HydraHarp 400, MultiHarp 150, MultiHarp 150 4N
- 3D piezo scanning
- Exit port, fiber-coupled with NIR PMT
This setup enables correlated AFM–FLIM measurements and extends detection into the near-infrared, including phosphorescence lifetime imaging (PLIM).
The integration required independent optical access, flexible detector placement, and adaptable TCSPC electronics. The AFM and the time-resolved confocal microscope operate as a coordinated system without compromising timing precision.

Spectrally and Temporally Resolved Multiphoton Measurements
Cristian Strassert – Center for Nanotechnology, WWU Münster, Germany
Research on photoactive coordination compounds often requires simultaneous spectral and temporal resolution, combined with single- and multi-photon excitation.
In this configuration, the MicroTime 200 (microscope body: Olympus IX73) was coupled to:
- A Spectra Physics MAI TAI multiphoton laser with APE double pulse picker
- An Andor SR163 spectrograph
- The FluoTime 300 High-End Photoluminescence Spectrometer
- The MultiHarp 150 Multichannel Time Tagging & TCSPC Unit
- A Excelitas SPAD and a PMA Hybrid-40 Photomultiplier Detector
The FluoTime 300 can operate independently or as an integrated extension of the microscope. This allows spatially resolved lifetime imaging (TRPL) at the microscope and complementary time-resolved emission scans using the spectrometer.

Cryogenic Single-Molecule Detection with Multi-Channel Timing
Gilad Haran – Weizmann Institute of Science, Israel
Single-molecule spectroscopy under cryogenic conditions imposes strict constraints on optical access, detector configuration, and mechanical integration. In addition to high timing precision, the setup must allow spectrally and polarization-resolved detection while operating in parallel with a cryostat.
In this configuration, the system included:
- Excitation at 485, 560, 595, 690, and 730 nm
- Olympus IX83 microscope body
- 3D piezo scanning
- Parallel integration of an Attocube cryostat
- Four Excelitas SPAD detectors
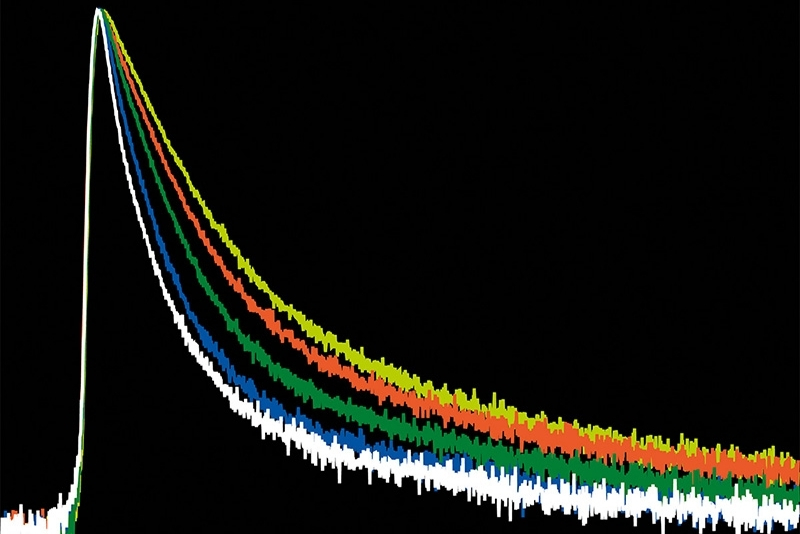
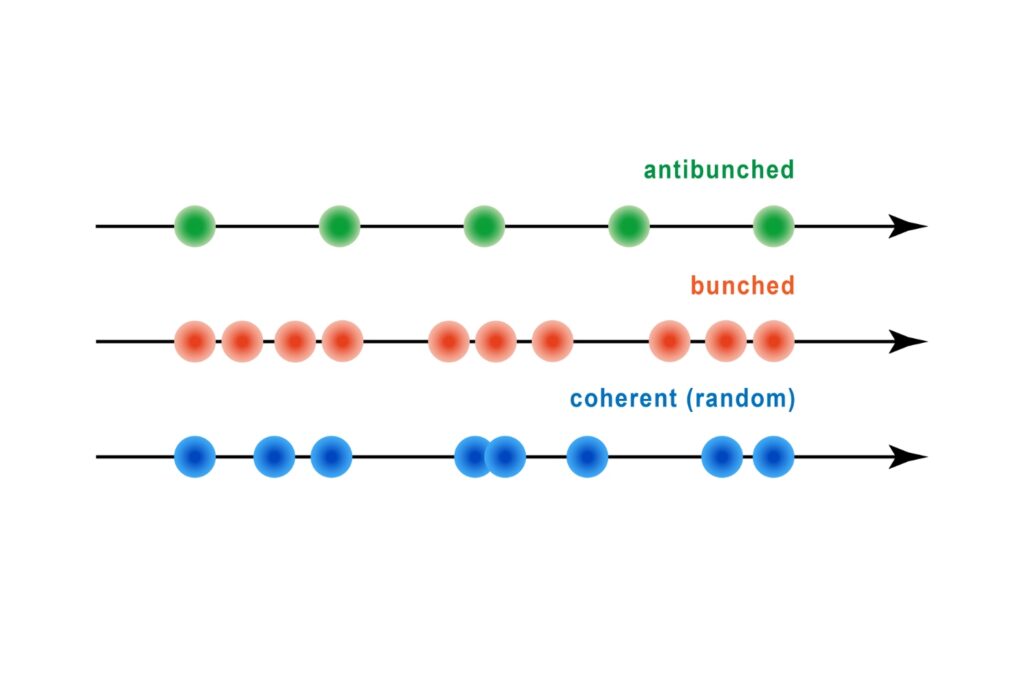
- Detection geometry enabling anisotropy and antibunching measurements
- Spectrograph receiving emission from the cryostat
- HydraHarp 400 Time Tagging & TCSPC Unit
The four-channel free-space detection geometry supports photon correlation measurements and time-resolved single-molecule spectroscopy under both cryogenic and room-temperature conditions.
The key requirement in this setup was detector flexibility combined with stable timing electronics. The cryostat operates in parallel to the confocal system, while timing synchronization remains consistent across channels.

Charge Carrier Diffusion Mapping in Emerging Semiconductors
Sam Stranks – University of Cambridge, Cavendish Laboratory, UK
In semiconductor research, spatially resolved charge carrier dynamics provide insight into recombination processes, defect densities, and diffusion behavior.
To investigate these properties, the system was configured with:
- Excitation at 405, 510, and 640 nm
- Multiphoton excitation using an Insight DS+ laser
- Olympus IX73 microscope body modified for illumination through the back port
- Cryostat integration
- FLIMbee galvo scanner
- Excelitas SPAD and PMA Hybrid-42 detectors
- Fiber-coupled Andor Kymera 193 spectrograph
- Time Tagging & TCSPC Electronics: A TimeHarp 260 Pico and TimeHarp 260 Nano
A defining feature of this configuration is the separation of excitation and detection volumes. By exciting locally through the back port and scanning the detection volume, spatially resolved “diffusion maps” can be recorded.
This architecture enables time-resolved imaging of carrier diffusion under both cryogenic and room-temperature conditions, without redefining the core detection or timing framework.

Defining Your Own Experimental Architecture
The four configurations described above differ significantly in scientific focus and technical implementation. They illustrate a system architecture in which excitation, detection, scanning, and timing electronics can be defined independently. This enables researchers to configure the instrument according to experimental requirements rather than adapting their measurement strategy to a fixed hardware structure.
When planning a new installation or expanding an existing setup, key parameters typically include wavelength coverage, detector configuration, spectral resolution, integration of cryogenic environments, or correlation with external instruments such as spectrometers. A modular time-resolved confocal microscope must accommodate these requirements while maintaining timing precision and detection performance.
Discuss your experimental requirements with our scientific team to define a customized MicroTime 200 configuration tailored to your laboratory.