Key Highlights
- Two-color 3D MINFLUX enables nanometer-precision tracking of single transport complexes inside transport-active nuclear pore complexes.
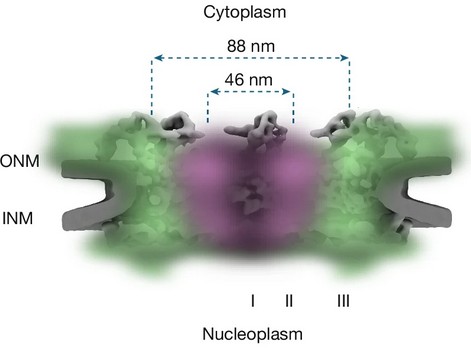
- Nuclear import and export occur within the same ~46 nm diameter annulus of the central pore channel.
- Translocation is highly confined and ~1,000-fold slower than free diffusion in solution.
- Radial density analysis reveals a central non-binding zone and peripheral high-affinity regions for transport receptors.
The Throughput Paradox of the Nuclear Pore
The nuclear pore complex (NPC) mediates one of the most demanding transport processes in the cell. Thousands of macromolecules per second pass through a channel only ~50–70 nm wide, moving simultaneously in opposite directions. From a physical perspective, this raises an immediate question: how does such intense bidirectional traffic avoid congestion and collision?
One long-standing hypothesis proposed spatially separated routes for nuclear import and export. However, directly visualizing millisecond transport events with nanometer precision inside intact, transport-active NPCs has remained technically challenging. Limited photon budgets, rapid photobleaching, and insufficient temporal resolution have historically restricted the number of localizations obtainable within the pore itself.
The study by Sau et al., Nature (2025), overcame these limitations using two-color 3D MINFLUX tracking.
What Two-Color 3D MINFLUX Revealed
By combining high spatiotemporal precision with reduced photobleaching, the authors were able to track single importin α complexes during both import and export through individual NPCs.
Several key observations emerged:
- Nuclear import and export do not occur in separate channels. Instead, both processes occupy the same ~46 nm diameter annular region within the central pore.
- Transport trajectories were largely excluded from the central axis of the pore.
- Movement inside the pore was highly confined and frequently interrupted by transient pauses.
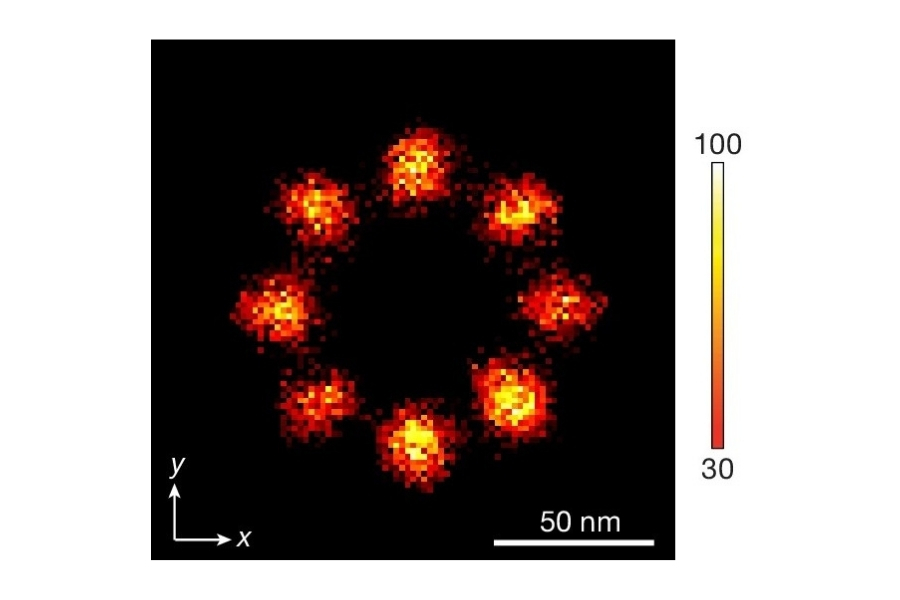
- Transport paths were often restricted to individual octants of the eightfold symmetric NPC scaffold.
Rather than a simple open conduit, the NPC permeability barrier appears structurally organized and functionally compartmentalized.
A Three-Zone Model of the Permeability Barrier
Radial density analysis revealed a spatially stratified architecture within the pore:
- Central non-binding zone: A region near the central axis largely devoid of transport receptor occupancy.
- Transport-active annulus (~23–30 nm radius): The zone in which active import and export translocation occurs.
- Peripheral high-affinity binding region: Areas near the pore wall exhibiting strong binding of transport receptors and translocation-arrested complexes.
This annular transport model revises earlier assumptions about central transport pathways and provides a structural framework for understanding bidirectional trafficking without requiring physically separate routes.
Confinement Quantified: Why Diffusion Measurements Matter
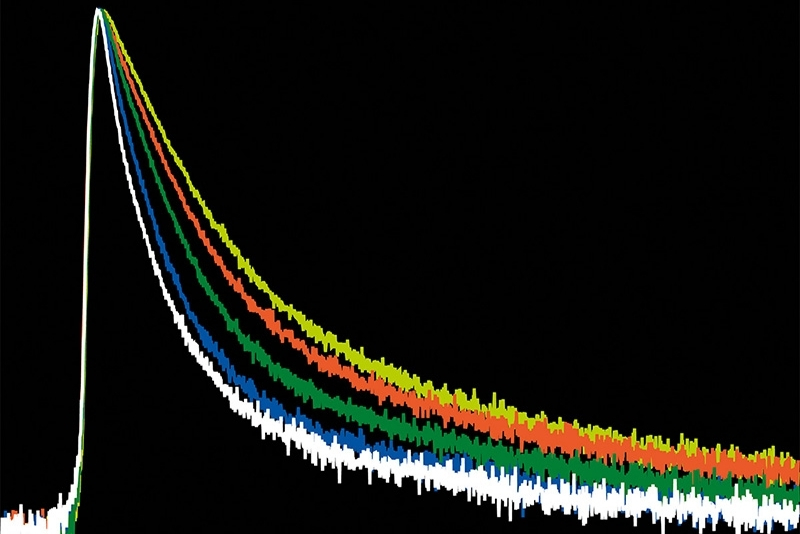
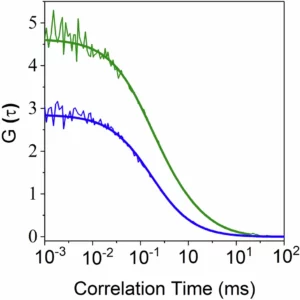
Spatial mapping alone does not fully describe transport. The physical constraints inside the pore must also be quantified. MINFLUX tracking yielded an apparent diffusion coefficient of approximately D ≈ 0.055 µm²/s inside the NPC. For comparison, fluorescence correlation spectroscopy (FCS) measurements of the same transport complexes in solution revealed diffusion coefficients between 14 and 64 µm²/s in buffer conditions. This corresponds to an approximately 1,000-fold reduction in mobility inside the pore.
Such a dramatic slowdown indicates that translocation occurs in an environment comparable to highly viscous media. The permeability barrier does not merely filter molecules by size. It also imposes substantial dynamic constraints on receptor–cargo complexes.
Instrumentation Used in This Study by PicoQuant
Luminosa Single Photon Counting Confocal Microscope

In this study, the Luminosa system was used for:
- Confocal imaging of NPC-bound transport receptors
- Fluorescence correlation spectroscopy (FCS) measurements
- Quantitative determination of diffusion coefficients in solution
- Characterization of transport complex diffusion behavior outside the pore
Key technical characteristics relevant to this work include:
- Single-photon counting detection for high sensitivity
- Integrated FCS module for diffusion analysis
- Pulsed excitation capability for time-resolved measurements
- Stable confocal alignment for quantitative fluorescence mapping
Alongside MINFLUX tracking of intrapore trajectories, confocal imaging and fluorescence correlation spectroscopy provided quantitative diffusion measurements that defined the mobility baseline outside the pore.
Implications for Quantitative Cellular Transport Research
The findings challenge the notion of centrally segregated trafficking routes and instead support an annular transport model with spatially differentiated binding zones.
More broadly, the work demonstrates that:
- Intracellular transport is far from free diffusion.
- Structural organization within nanoscale channels imposes measurable kinetic constraints.
- Mechanistic conclusions require both spatial precision and quantitative diffusion analysis.
This study illustrates how spatially resolved single-molecule tracking and quantitative diffusion analysis together enable a mechanistic interpretation of nucleocytoplasmic transport.
Explore how Luminosa enables quantitative fluorescence correlation spectroscopy for diffusion measurements in complex biological systems.