Photon Counting Detectors


Simplify your materials characterization with one flexible TRPL microscope enabling multiple methods for precise and efficient analysis.

Complete confocal fluorescence microscope that empowers researchers to advance quantitative functional imaging from individual molecules to cells and tissues.

Compact FLIM and FCS upgrade kit that adds advanced functional imaging and correlation analysis to existing laser scanning microscopes.

Designed for flexible, sensitive, and precise steady-state and time-resolved spectroscopy across the UV to NIR range and time scales from picoseconds to milliseconds.

Modular lifetime spectrometer designed for flexible fluorescence and photoluminescence measurements in both materials and life science research.

Add spectral and time-resolved photoluminescence to your setup through flexible microscope–spectrometer coupling options.

Get the most out of superconducting nanowire detectors in large-scale quantum communication and computing experiments requiring precise multichannel timing.

Boost your time-resolved experiments with a flexible, high-precision time tagging and TCSPC unit for materials science and quantum sensing.

Scale your photonic quantum computing and detector characterization setups while maintaining performance, flexibility, and high data throughput.

Compact 3-color picosecond laser delivering flexible ns to ms excitation with cost-effective multicolor performance and straightforward operation.

Smart picosecond laser diode heads covering UV-A to NIR, providing the right combination of power, pulse width, and diode type for any time-resolved technique.

VisUV provides clean short pulses and stable timing across key UV and visible wavelengths, including deep UV lines as well as 488 nm and 532 nm.

Enhance your single-photon counting experiments with wide dynamic range and excellent timing precision in the UV and visible even at the highest count rates.

Capture even the weakest signals over large areas with maximum dynamic range and enhanced low-light sensitivity in a compact detector design.

Unlock spatially resolved single-photon detection with a 23-pixel SPAD array, combining low dark counts and precise time tagging for advanced experiments.
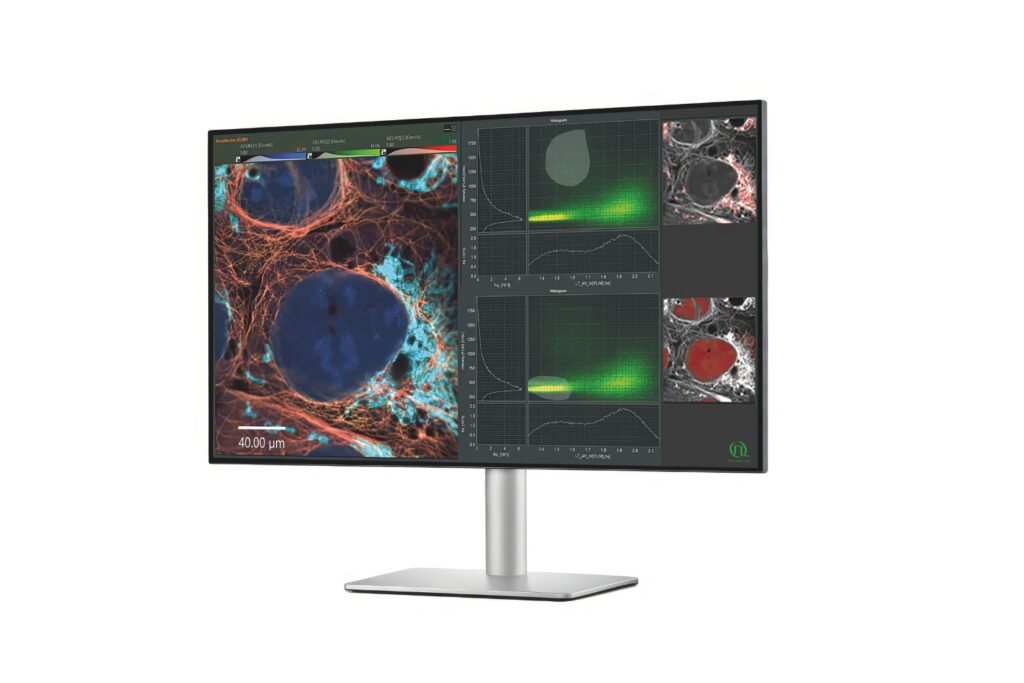
Advanced FLIM analysis software for fast, accurate interpretation of lifetime imaging data.
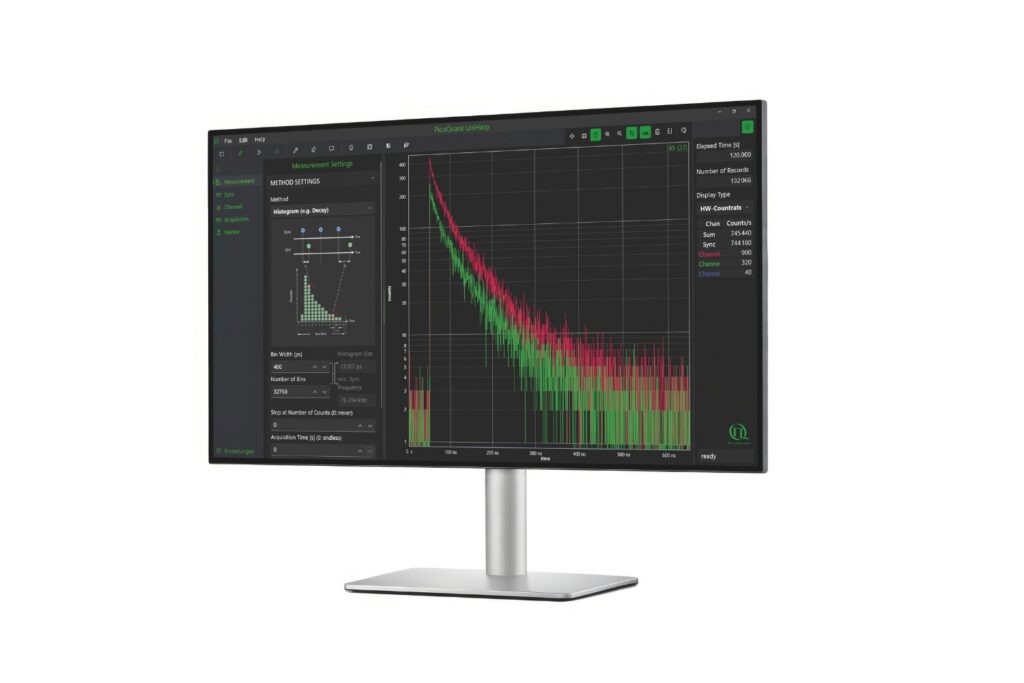
Intuitive, free software solution for real-time, high-precision photon data acquisition, visualization, and initial data analysis.
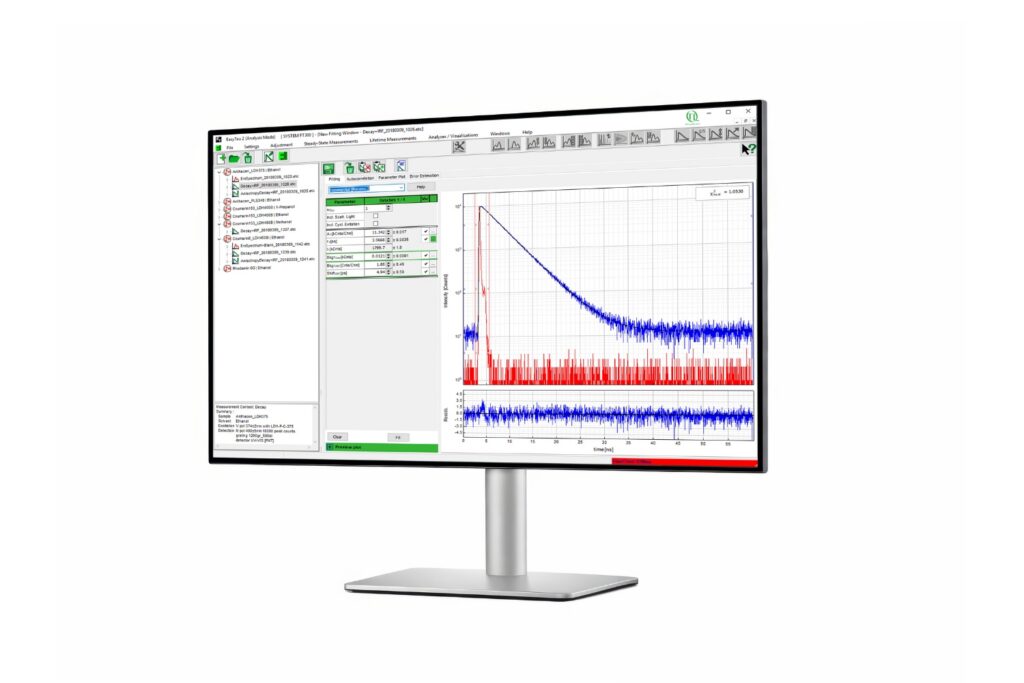
Advanced software for time-resolved fluorescence acquisition and analysis.

An imaging technique that uses fluorescence lifetimes to generate image contrast.
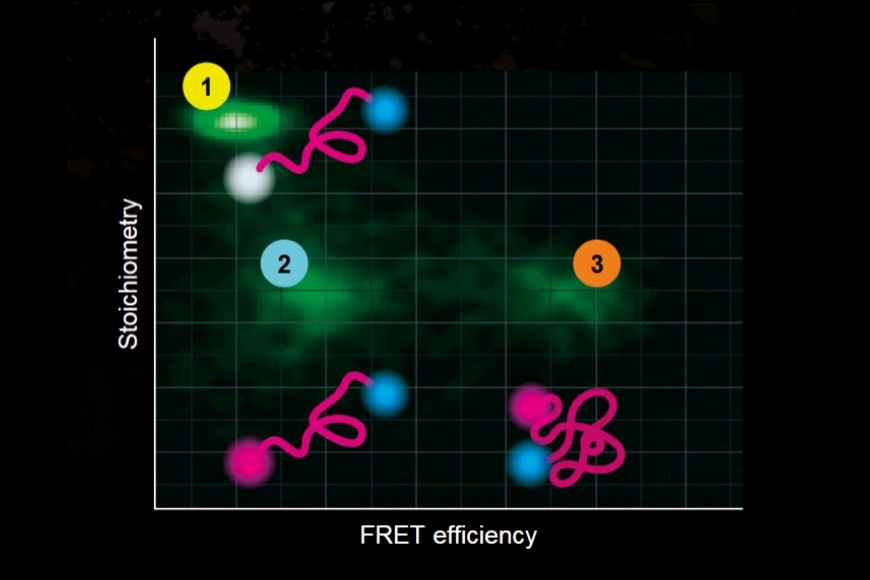
Investigating how proteins dynamically explore multiple conformational states that control biological function.

Investigating how biomolecules separate into dynamic liquid phases to organize cellular space and regulate biological function.
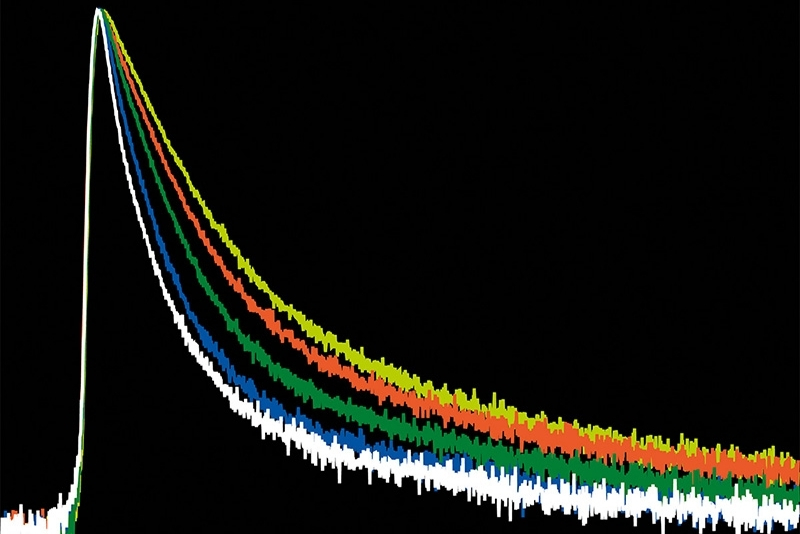
A time-resolved technique that measures photoluminescence lifetimes to reveal excited-state dynamics in materials.
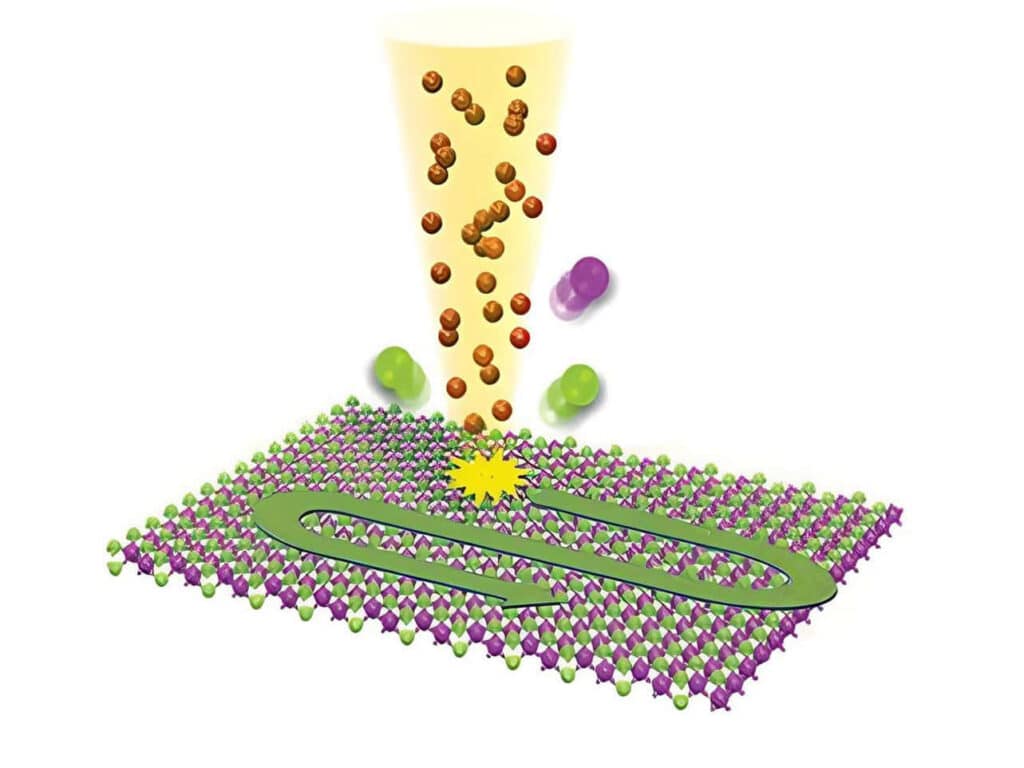
Studying exciton dynamics, charge carrier processes, and structural properties through optical and time-resolved characterization methods.

Investigating charge-carrier lifetimes and recombination dynamics to enable precise optical characterization of material quality and device performance.
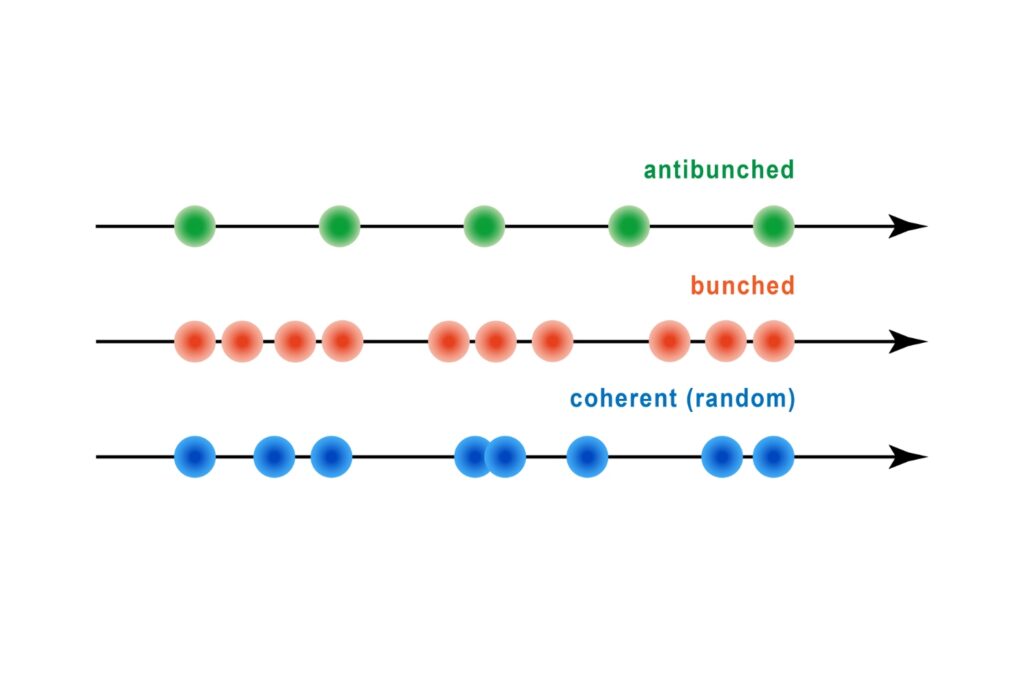
A quantum optical signature revealed by time-resolved photon correlation analysis to identify single-photon emission in materials and nanostructures.
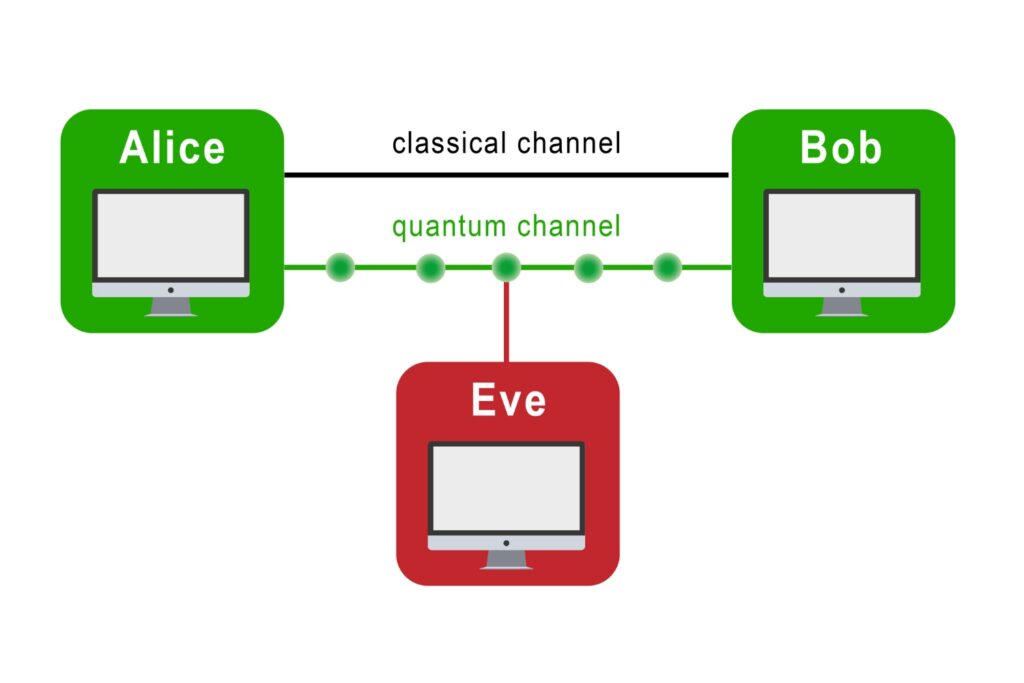
The transmission of information using individual photons, using quantum effects to ensure absolute security.
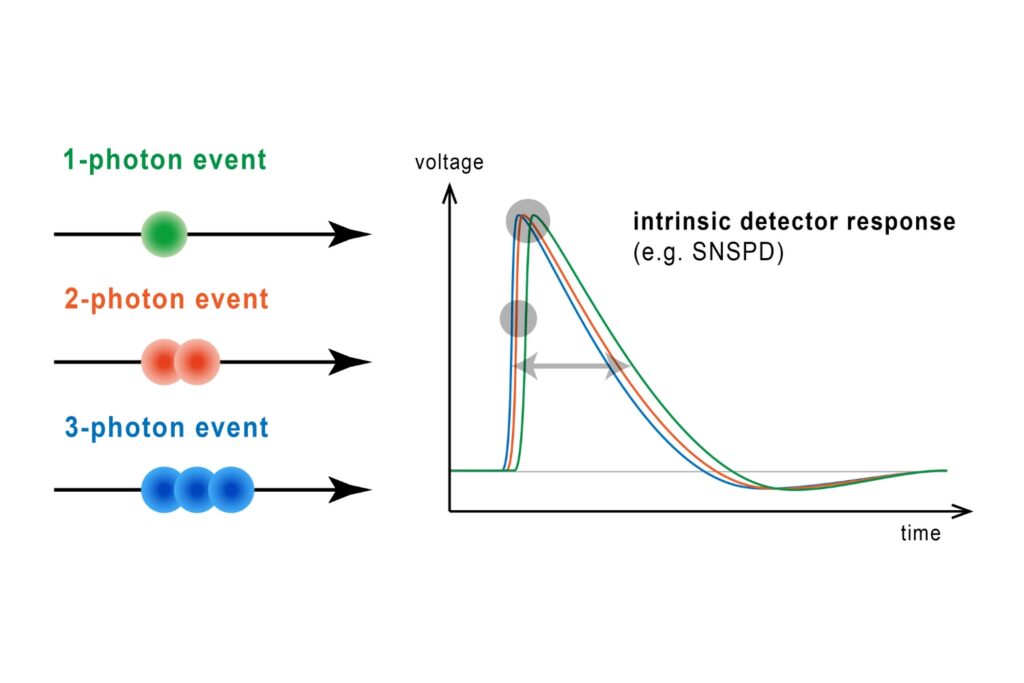
Quantifying photons per detection event enables direct access to photon-number statistics, providing insight into quantum and statistical properties of light.

An optical technique that analyzes light emission under electrical excitation to reveal electronic properties of electroluminescent materials.
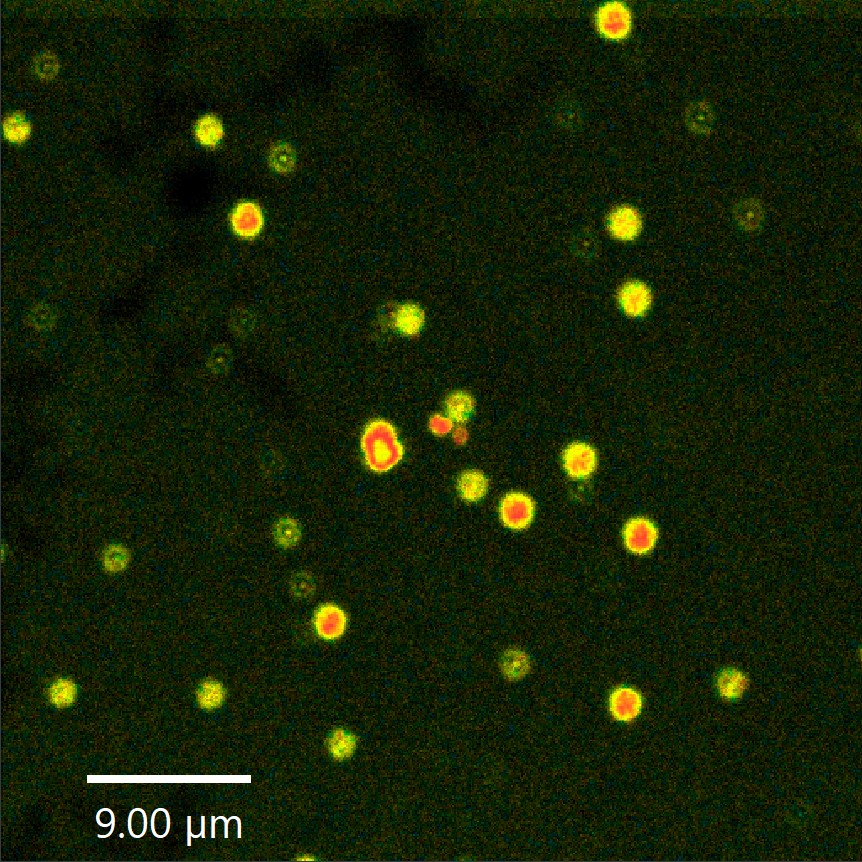
Monitoring environmental signals and trace compounds to understand dynamic changes in natural and engineered environments.
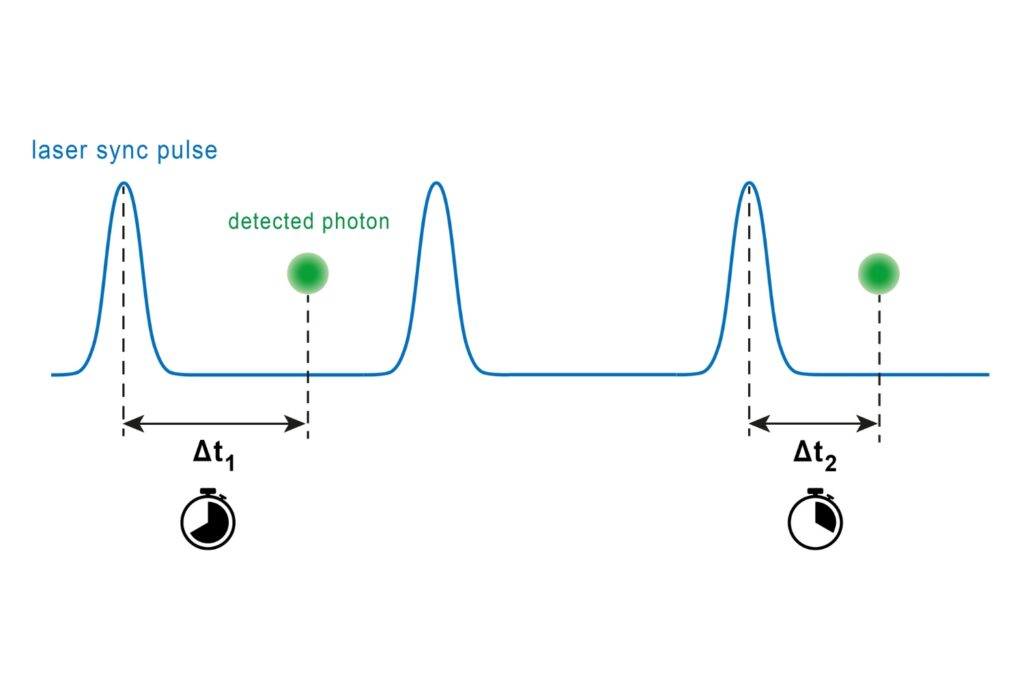
A photon timing technique that measures single-photon arrival times to resolve ultrafast dynamics in fluorescence, materials research, and quantum optics.

Liquid–liquid phase separation (LLPS) is a biophysical process in which a homogeneous solution of biomolecules, typically proteins, nucleic acids, or their complexes, spontaneously separates into two coexisting liquid phases. One phase becomes enriched in specific biomolecules and forms condensates, while the other remains a dilute surrounding phase.
This separation occurs without membranes and gives rise to dynamic, reversible, and functionally specialized compartments known as biomolecular condensates or membraneless organelles. LLPS can be triggered by changes in concentration, temperature, pH, or ionic strength, leading to the formation of cellular structures such as stress granules, P-bodies, and nucleoli that selectively concentrate proteins and RNAs while maintaining liquid-like dynamics.
The formation, dissolution, and material state transitions of these condensates are governed by a finely tuned balance of multivalent molecular interactions, intrinsically disordered proteins (IDPs), intrinsically disordered regions (IDRs), and environmental conditions.
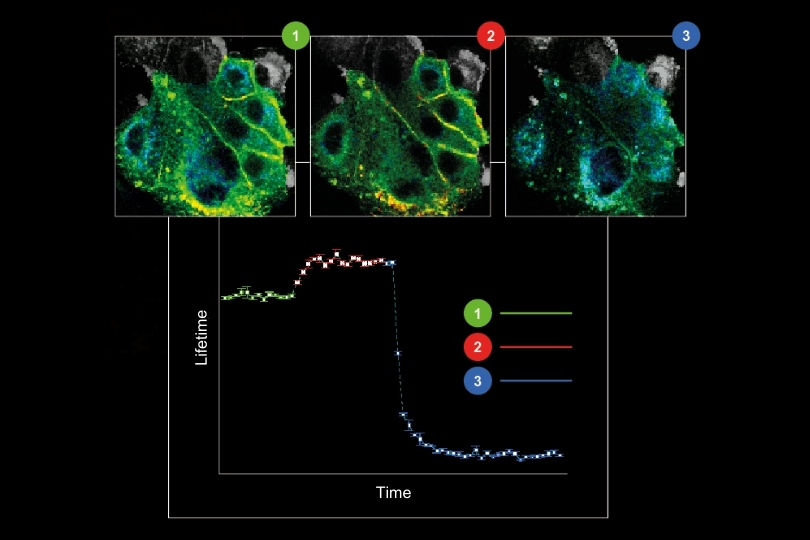
 Fluorescence lifetime imaging microscopy highlights spatial variations within biomolecular condensates, reflecting their dynamic organization and functional diversity in LLPS.
Fluorescence lifetime imaging microscopy highlights spatial variations within biomolecular condensates, reflecting their dynamic organization and functional diversity in LLPS.Understanding liquid–liquid phase separation (LLPS) is key to uncovering how cells organize their biochemistry without membranes. LLPS provides a conceptual framework for how biomolecular condensates regulate essential processes such as RNA metabolism in stress granules, ribosome biogenesis in the nucleolus, and chromatin organization during DNA repair. Because LLPS is reversible and tightly controlled, it allows cells to assemble or disassemble functional compartments in response to environmental cues. When these phase transitions become dysregulated, they can lead to pathological states including the protein aggregation seen in amyotrophic lateral sclerosis (ALS), frontotemporal dementia (FTD), and certain cancers. Studying LLPS therefore reveals the principles underlying cellular organization and disease.
Fluorescence-based techniques are particularly well suited to studying liquid–liquid phase separation, as they enable quantitative analysis of molecular dynamics, interactions, and local environments in both dilute and condensed phases. Fluorescence correlation spectroscopy (FCS) provides access to molecular diffusion and exchange between phases, revealing differences in mobility inside and outside biomolecular condensates. Förster resonance energy transfer (FRET) and fluorescence anisotropy probe molecular interactions and local crowding within condensates, while fluorescence lifetime measurements and fluorescence lifetime imaging microscopy (FLIM) add sensitivity to environmental changes and heterogeneity. Together, these complementary approaches allow LLPS to be characterized across multiple length and time scales under physiologically relevant conditions.
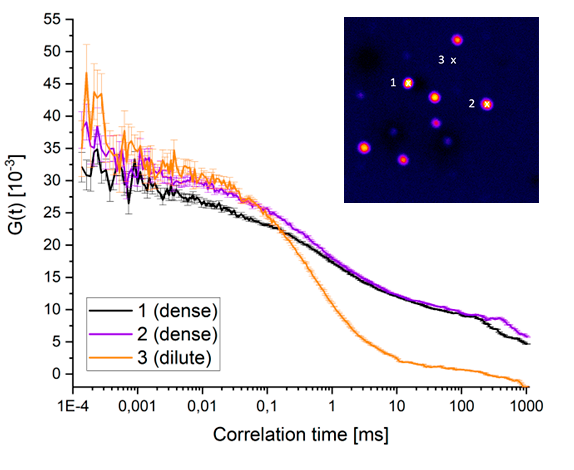 Fluorescence correlation spectroscopy reveals pronounced differences in molecular diffusion inside and outside biomolecular condensates, with slower diffusion in the dense phase indicating altered molecular mobility and heterogeneity.
Fluorescence correlation spectroscopy reveals pronounced differences in molecular diffusion inside and outside biomolecular condensates, with slower diffusion in the dense phase indicating altered molecular mobility and heterogeneity.Using synapsin-1 as a model system, phase-dependent differences in molecular mobility and heterogeneity can be resolved by combining FLIM and multi-point FCS. These measurements provide quantitative insight into how proteins dynamically partition and diffuse between dilute and condensed phases.
Using time-resolved fluorescence anisotropy and FRAP, researchers investigated the early stages of α-synuclein aggregation during liquid–liquid phase separation. Measurements inside and outside liquid droplets revealed reduced rotational mobility and increased molecular rigidity within the condensed phase. The results showed that LLPS precedes α-synuclein aggregation and that liquid-like droplets progressively undergo a liquid-to-solid transition during ageing, providing mechanistic insight into aggregation pathways linked to Parkinson’s disease.
Using single-molecule fluorescence techniques, researchers investigated the conformational dynamics of the intrinsically disordered SARS-CoV-2 nucleocapsid protein and its ability to undergo liquid–liquid phase separation with RNA. By combining smFRET and nanosecond FCS, distinct conformational subpopulations and rapid chain reconfiguration dynamics were resolved. The results revealed how intrinsic disorder and molecular flexibility contribute to phase separation and viral genome packaging, providing mechanistic insight into N protein function.

This application note shows how Luminosa enables FLIM and multi-point FCS to study liquid–liquid phase separation and protein dynamics at the single-molecule level.
Please fill out the form below to receive the requested file. After submitting your details, the file will be sent to you by email.
* Required
Please fill out the form below to request more information. You may also use it to inquire about pricing, availability, technical specifications, or discuss your specific application. Our sales team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information about our products and services. You may also use it to ask for pricing, availability, technical specifications, or any other details relevant to your inquiry. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information and prices about our product. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required