Key Highlights
- Fluorescence lifetime provides a concentration-independent parameter for robust cell classification in high-content screening workflows.
- Automated feedback microscopy enables dynamic cell selection based on statistically defined lifetime thresholds rather than intensity fluctuations.
- The LSM Upgrade Kit integrates TCSPC-based FLIM into existing laser scanning microscopes, enabling quantitative lifetime-driven screening without system replacement.
The Limitation of Intensity-Based Screening
High-content screening commonly relies on fluorescence intensity to classify cellular responses. Although intensity measurements are straightforward to implement, they remain sensitive to probe concentration, photobleaching, excitation fluctuations, and detector settings. In dynamic live-cell experiments, these variables can distort response amplitudes and complicate automated decision-making. Signal saturation further limits quantitative interpretation when studying processes such as calcium signaling. As screening workflows become increasingly automated, the dependence on intensity alone can reduce robustness and reproducibility across large cell populations.
Lifetime as a Quantitative Selection Parameter
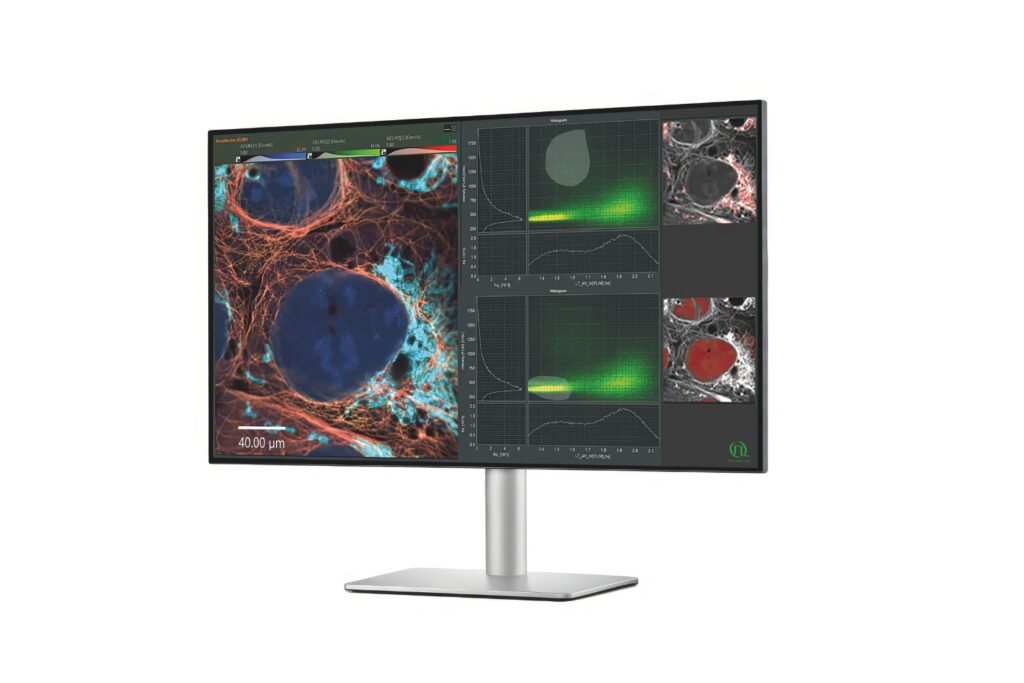
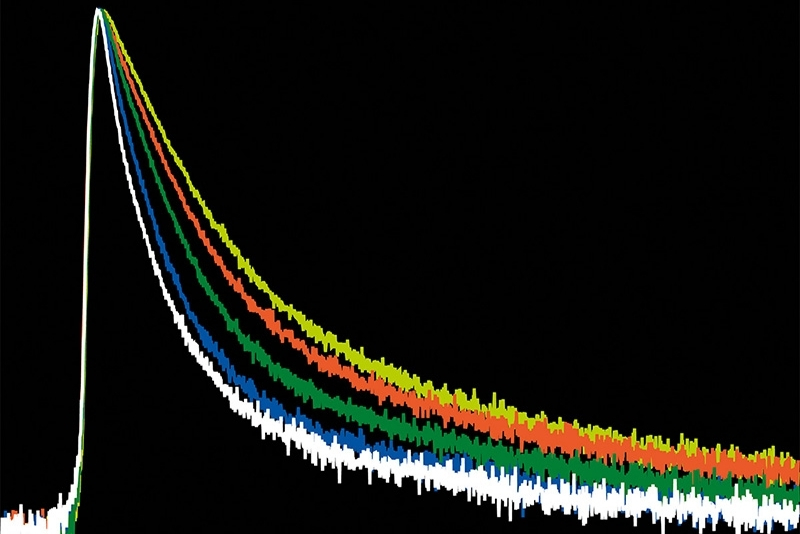
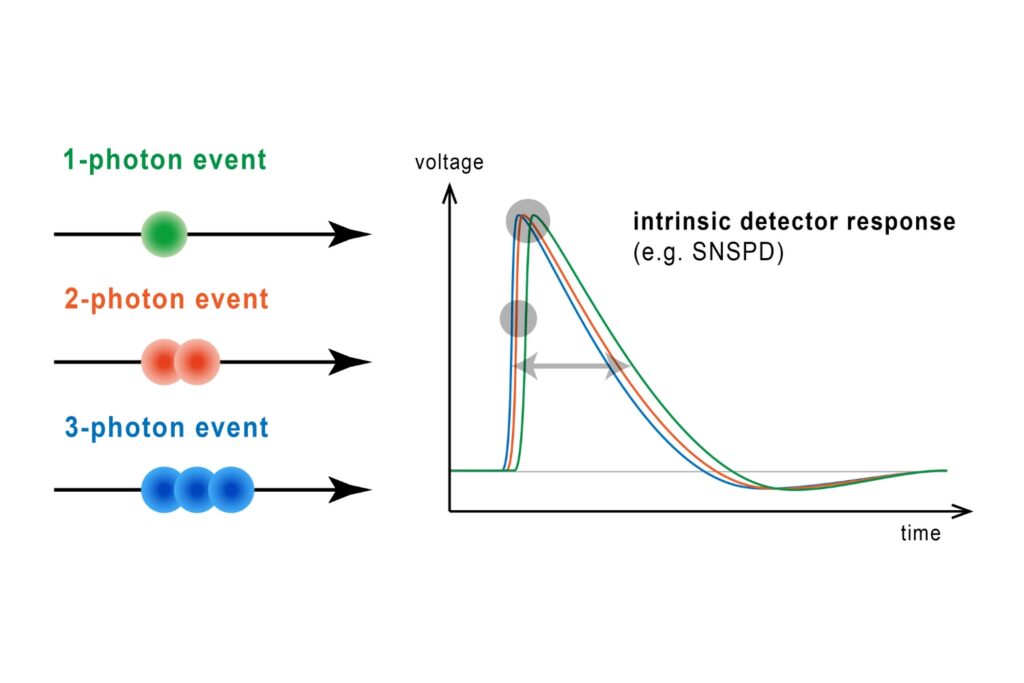
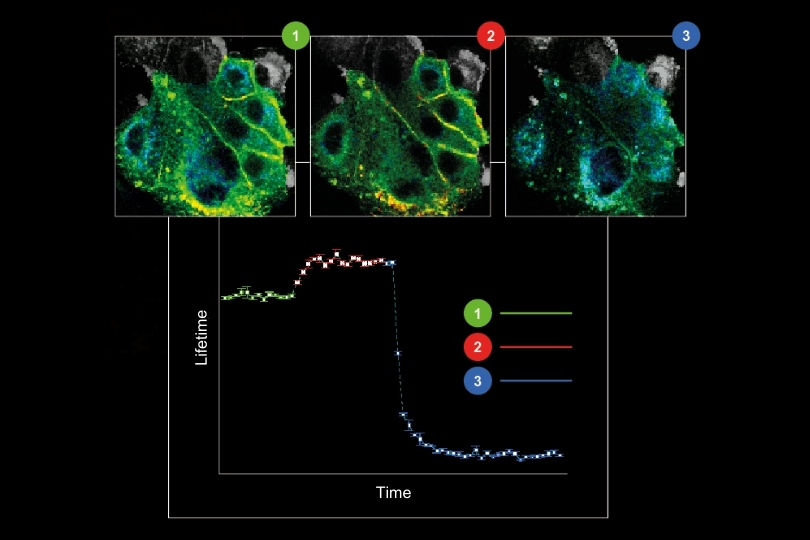
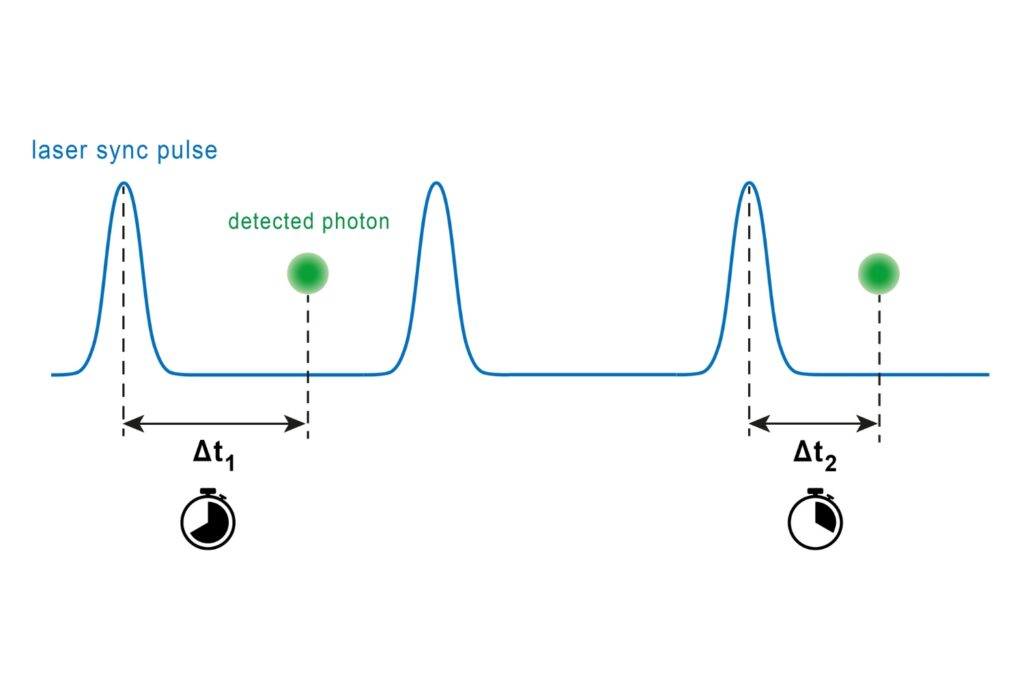
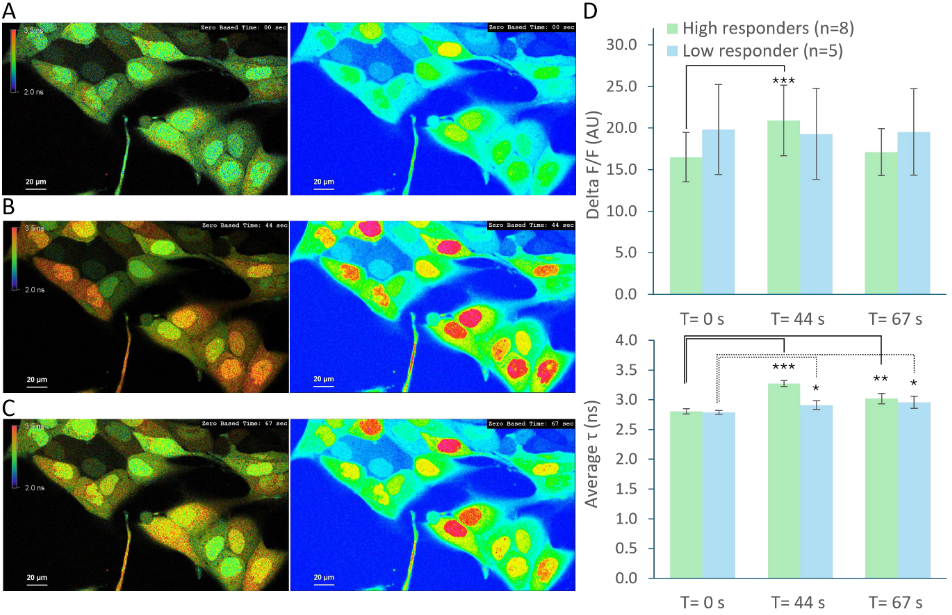
Fluorescence lifetime imaging microscopy (FLIM) provides an alternative parameter that is largely independent of fluorophore concentration and excitation power. In a feedback microscopy experiment on ATP-induced calcium signaling in MDCK cells loaded with Oregon Green BAPTA-1, presented in a Nikon application note using PicoQuant’s LSM Upgrade Kit, two response populations were identified. Intensity analysis clearly revealed strong responders but failed to distinguish subtle changes in a second group. Lifetime analysis, however, detected significant shifts in both populations. High responders showed a clear increase in average lifetime after ATP stimulation, while even cells with minimal intensity variation exhibited measurable lifetime changes. The smaller variability of lifetime measurements compared to intensity enabled more reliable discrimination between subpopulations. Phasor-based representation further allowed real-time visualization of the lifetime shift during stimulation, supporting rapid assessment of cellular responses.
From Imaging to Automated Feedback Microscopy
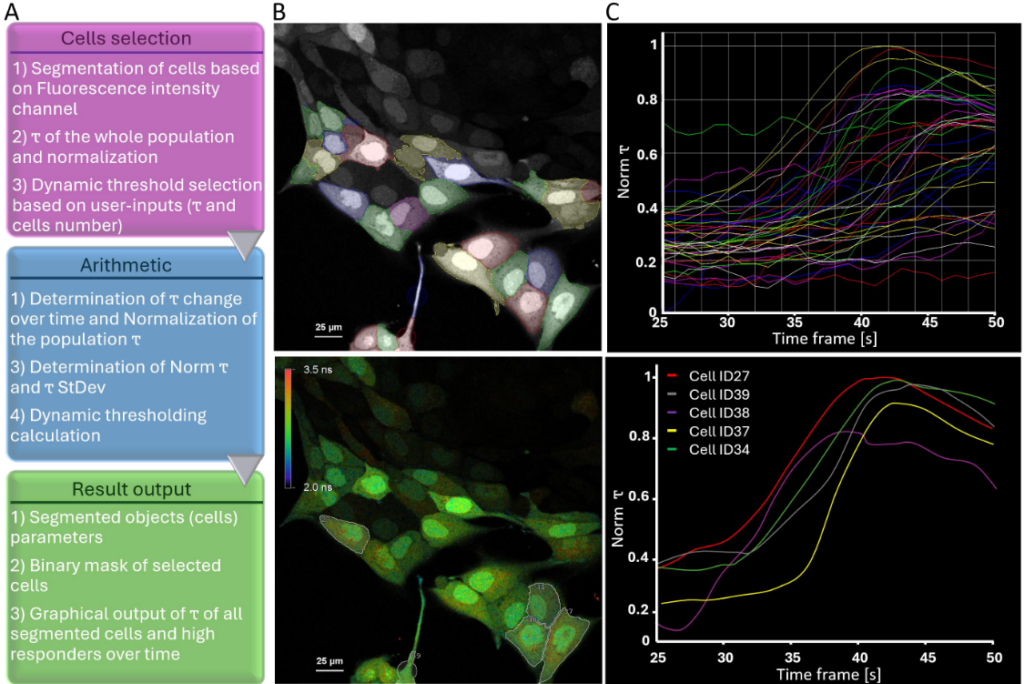
The workflow extended beyond image acquisition. Individual cells were segmented within the GA3 analysis environment of NIS-Elements, and lifetime dynamics were tracked over time. A dynamic threshold was calculated using the mean and standard deviation of lifetime changes across the population. This strategy enabled automatic identification of high responders without prior knowledge of response distribution. Artificial intelligence-assisted segmentation improved object detection in densely packed or low-resolution images. Once defined, the threshold criteria triggered a feedback loop in which selected cells could be re-imaged or subjected to additional microscopic tasks. Minimal user interaction was required after the experiment started, demonstrating how lifetime data can drive automated, quantitative screening workflows.
What This Means for Existing LSM Users
This study illustrates how time-resolved detection can transform a conventional laser scanning microscope into a quantitative screening platform. Rather than replacing established confocal systems, lifetime detection modules can be integrated into existing scan and detection pathways. This enables automated cell classification based on a physical decay parameter instead of intensity fluctuations. For facilities already operating laser scanning microscopes, the approach provides a practical route toward robust, lifetime-driven high-content experiments.
Instrumentation Used in This Study by PicoQuant
The experiment was performed using the PicoQuant LSM Upgrade Kit integrated into a Nikon AX R confocal microscope platform.
Key components included:
- Picosecond pulsed excitation at 485 nm
- Single-photon counting hybrid detectors
- MultiHarp 150 time tagging & TCSPC unit
- SymphoTime 64 data acquisition for quantitative lifetime analysis

Together, these components enabled robust fluorescence lifetime imaging and automated feedback microscopy based on quantitative decay parameters.