Key Highlights
- Reproducibility in fluorescence lifetime imaging microscopy (FLIM) depends on stable system architecture rather than post-acquisition correction.
- Quantitative FLIM accuracy is governed by photon statistics and limited by detector dead time under high count rate conditions.
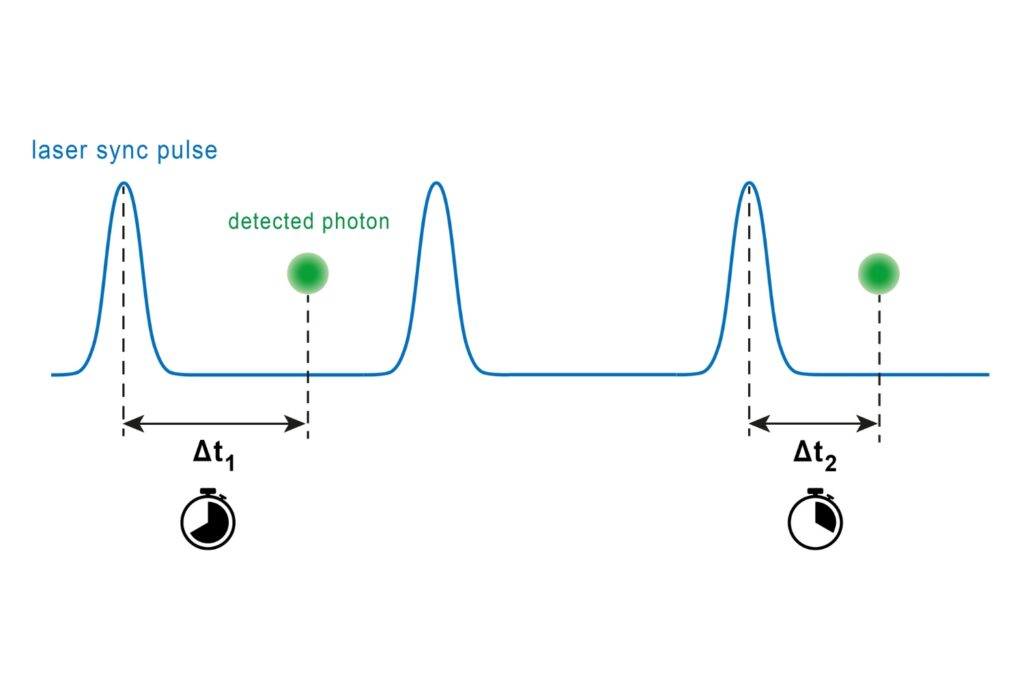
- In TCSPC-based FLIM, minimizing pile-up effects is essential to preserve decay fidelity.
- The Luminosa FLIM microscope integrates high-throughput photon-counting hardware with workflow-driven automation to support quantitative, reproducible imaging.
This article is based on the work by Maria Loidolt-Krueger published in Microscopy Today (2023).
Why Reproducibility Is a Persistent Challenge in Fluorescence Lifetime Imaging Microscopy
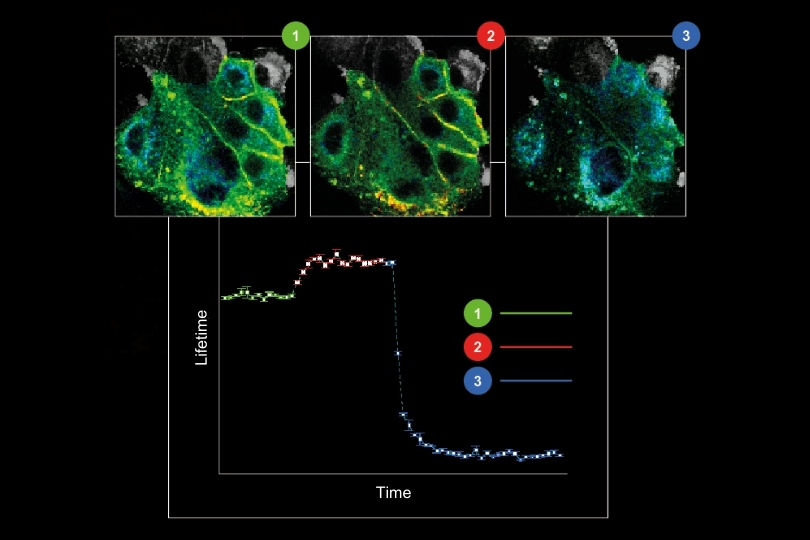
Fluorescence lifetime imaging microscopy (FLIM) offers concentration-independent contrast, yet quantitative FLIM remains sensitive to acquisition variability. Small changes in excitation power influence photon yield and photobleaching. Detector configuration, repetition rate, and fitting constraints directly affect decay statistics.
While such effects may be negligible in qualitative imaging, they become critical when lifetimes are used to quantify FRET efficiency, molecular interactions, or environmental parameters. Reproducibility in fluorescence microscopy therefore depends not only on sample preparation, but on stable system architecture and controlled acquisition parameters.
Photon Statistics, Count Rates, and Detector Dead Time in Quantitative FLIM
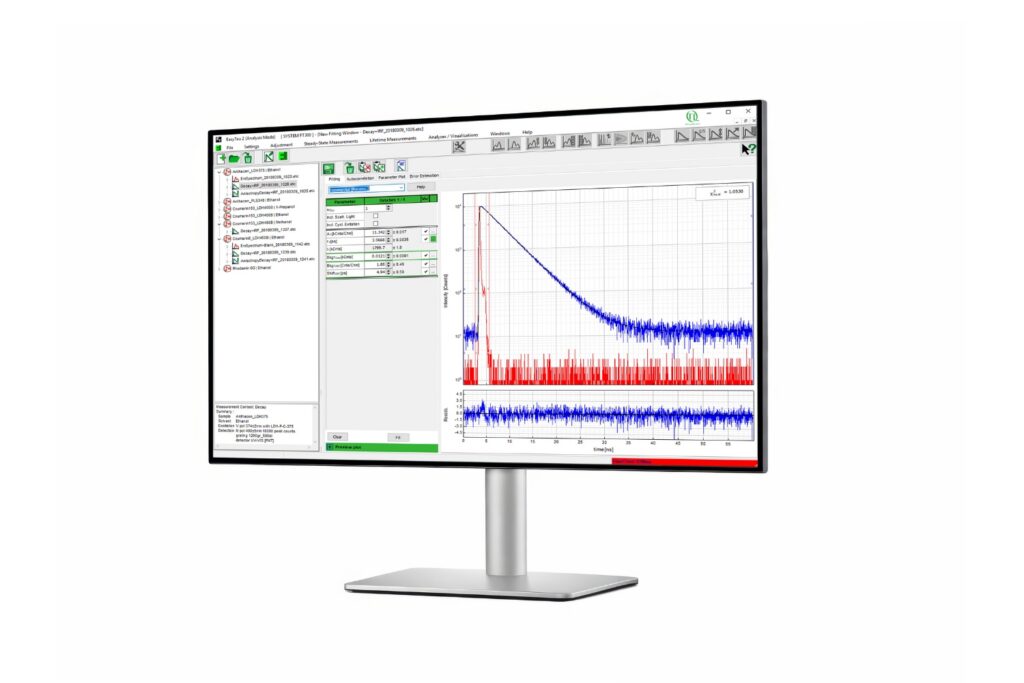
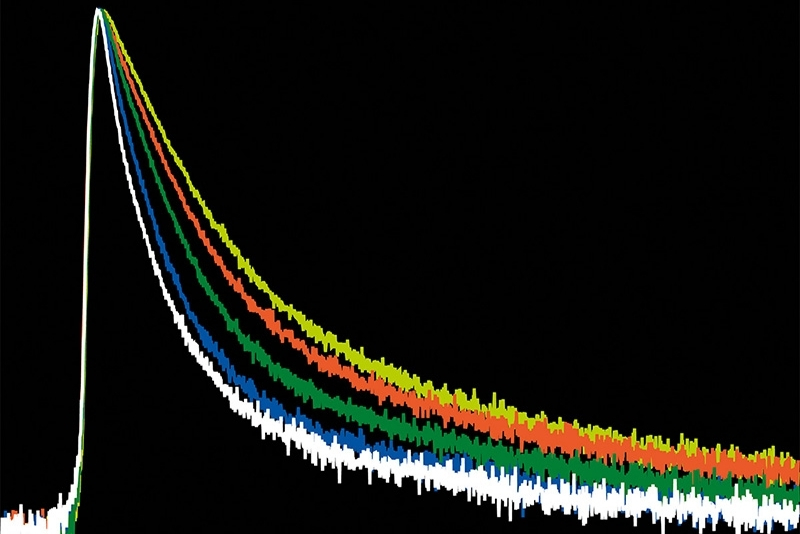
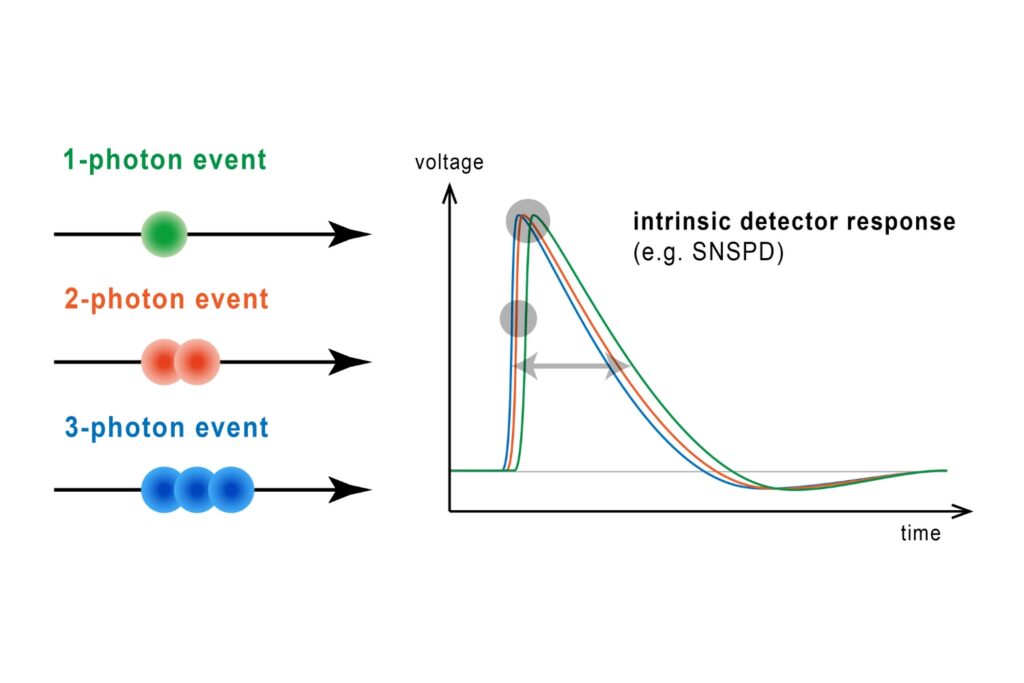
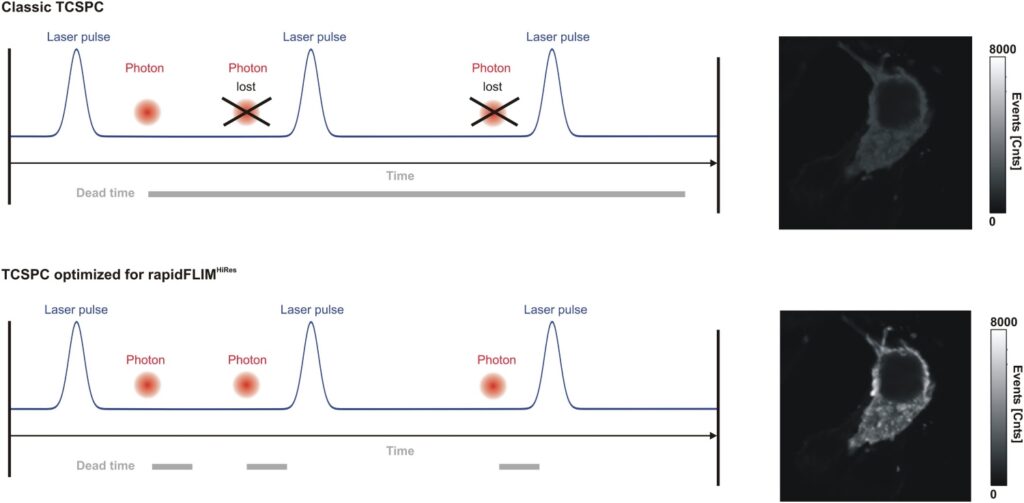
The precision of quantitative FLIM is governed by photon statistics. Lifetime accuracy improves with photon number, but high count rates introduce systematic distortions if detector dead time is not minimized.
In time-correlated single photon counting (TCSPC)-based FLIM, pile-up effects can artificially shorten measured lifetimes when detection electronics reach their recovery limit. A FLIM microscope for quantitative applications must therefore combine high sensitivity with short dead time and stable timing electronics to preserve decay fidelity under realistic imaging conditions.
Automation as a Design Principle for Reproducible FLIM Measurements
Variability introduced by manual system configuration remains a major limitation in lifetime imaging. Manual alignment, subjective power adjustment, and inconsistent acquisition criteria reduce comparability across experiments.
Automated alignment, calibrated excitation control, workflow-driven configuration, and metadata tracking transform reproducibility from a user responsibility into a system property, making . In quantitative FLIM, automation becomes a structural requirement for reliable measurements rather than a convenience feature.
The Luminosa FLIM Microscope Platform

A FLIM microscope designed for quantitative applications must preserve photon statistics at high count rates, minimize dead time distortion, and maintain stable excitation conditions across experiments. The Luminosa Single Photon Counting Confocal Microscope was designed to address these requirements at the architectural level.
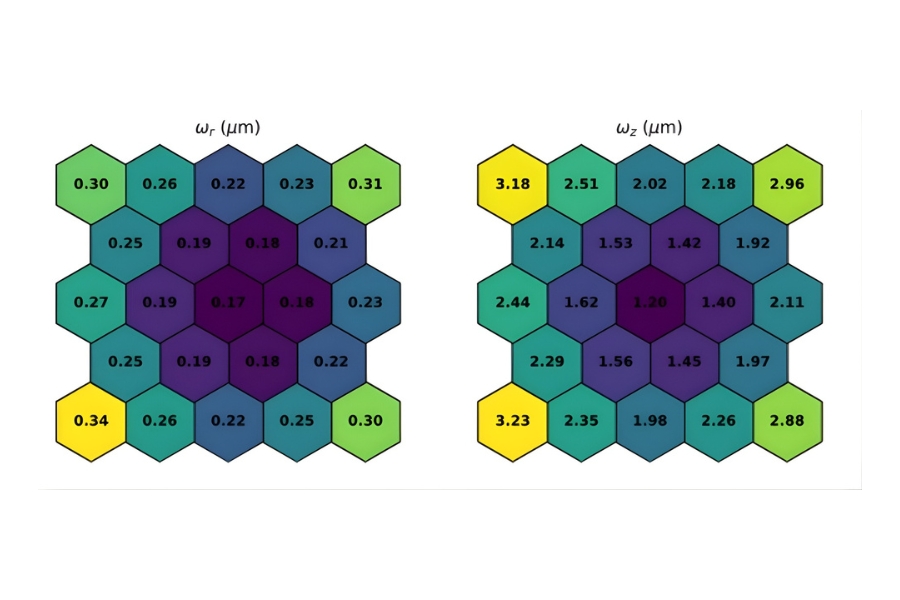
Its optical layout is optimized for sensitivity through a minimal number of high-quality optical elements. Users can switch between high-speed galvo scanning and piezo objective scanning. By bypassing scanning mirrors, piezo operation reduces photon loss and can increase detected signal by approximately 20 to 30 percent, depending on wavelength. For quantitative FLIM, improved photon yield directly enhances lifetime precision while limiting excitation power.
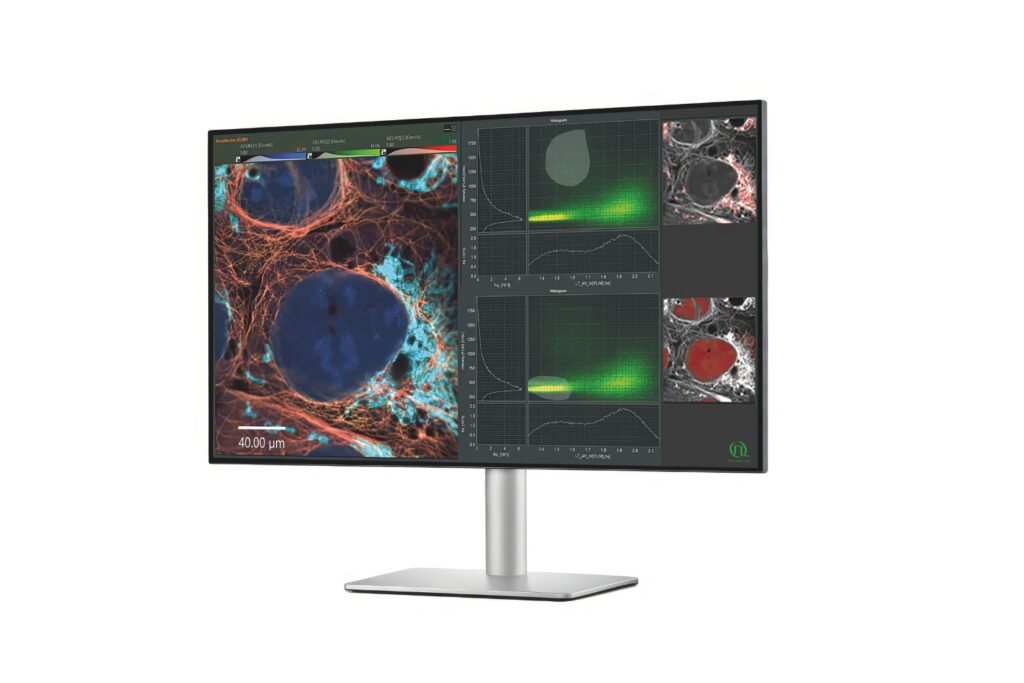
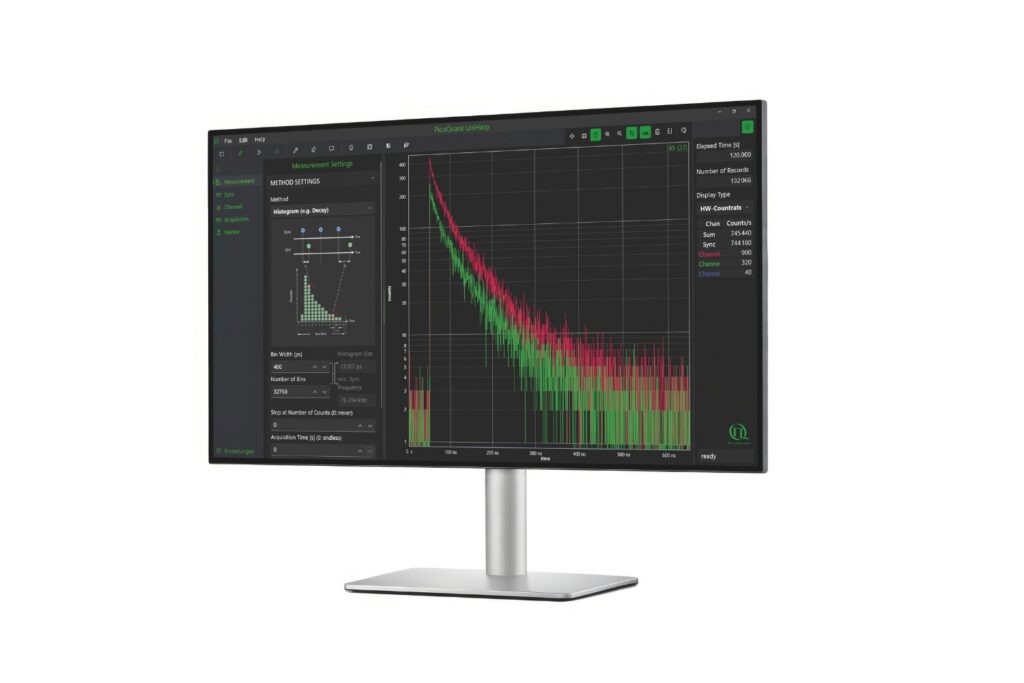
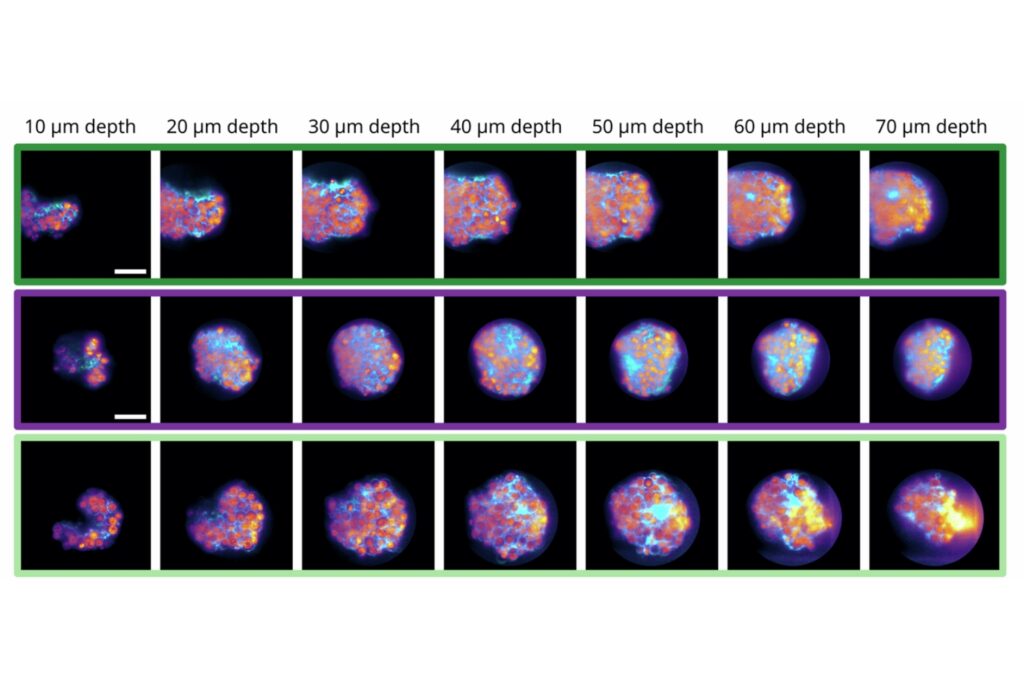
Luminosa integrates PMA Hybrid Photomultiplier Detectors with MultiHarp High-Throughput Multichannel Time Tagging & TCSPC electronics featuring an ultra-short dead time of 650 picoseconds. Sustained count rates of up to 78 Mcps enable rapidFLIMHiRes acquisition and frame rates of up to 15 FLIM images per second, depending on experimental conditions. High count rate capability supports accurate decay reconstruction under realistic imaging regimes.
Workflow-driven control further strengthens reproducibility in fluorescence microscopy. Sample-free auto-alignment, excitation power calibration in absolute units, automatic hardware configuration, defined stop criteria based on photon statistics, and real-time lifetime analysis reduce operator-dependent variability. GPU-accelerated processing analyzes a 1024 by 1024 pixel FLIM image in approximately five seconds while preserving raw data integrity.
These design choices align Luminosa with the requirements of quantitative FLIM and reproducible data acquisition.
Designing for Quantitative Reproducibility
Reproducibility in fluorescence microscopy is not achieved by post-processing alone. It must be embedded in system design. By combining optimized photon-counting hardware with automation-driven configuration and calibration, modern FLIM microscope platforms such as Luminosa support reliable, quantitative imaging.