Key Highlights
- Quantitative FLIM relies on accurate photon timing and faithful decay reconstruction rather than intensity alone.
- High count rate FLIM improves statistical precision but introduces dead time and photon pile-up effects that can distort measured lifetimes.
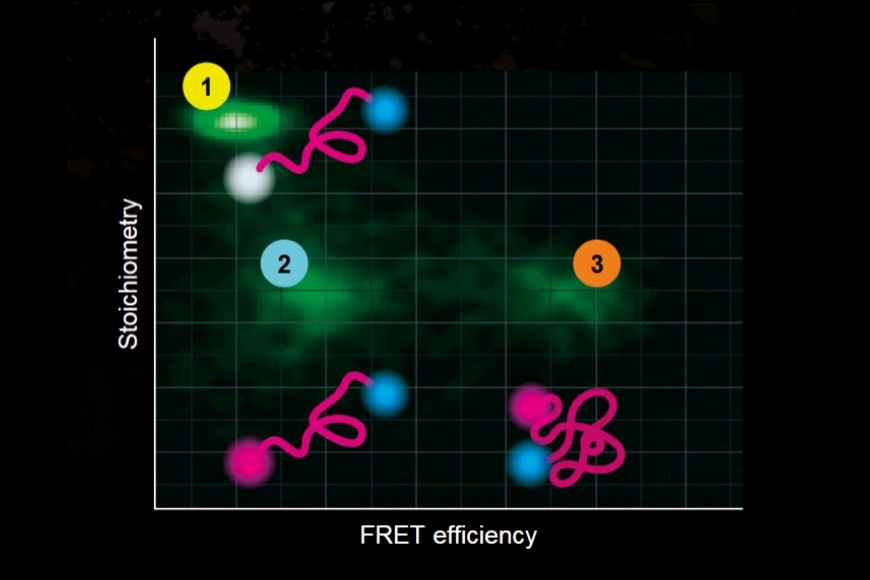
- In FLIM-FRET and single-molecule FRET measurements, even small decay distortions directly affect extracted energy transfer efficiencies and distance estimations.
- A FLIM microscope designed for sustained count rate handling and short detector dead time preserves lifetime accuracy under elevated photon flux.
This article builds on the work published by Maria Loidolt-Krueger in Microscopy Today (2023).
Single-Photon Counting as the Foundation of Quantitative FLIM
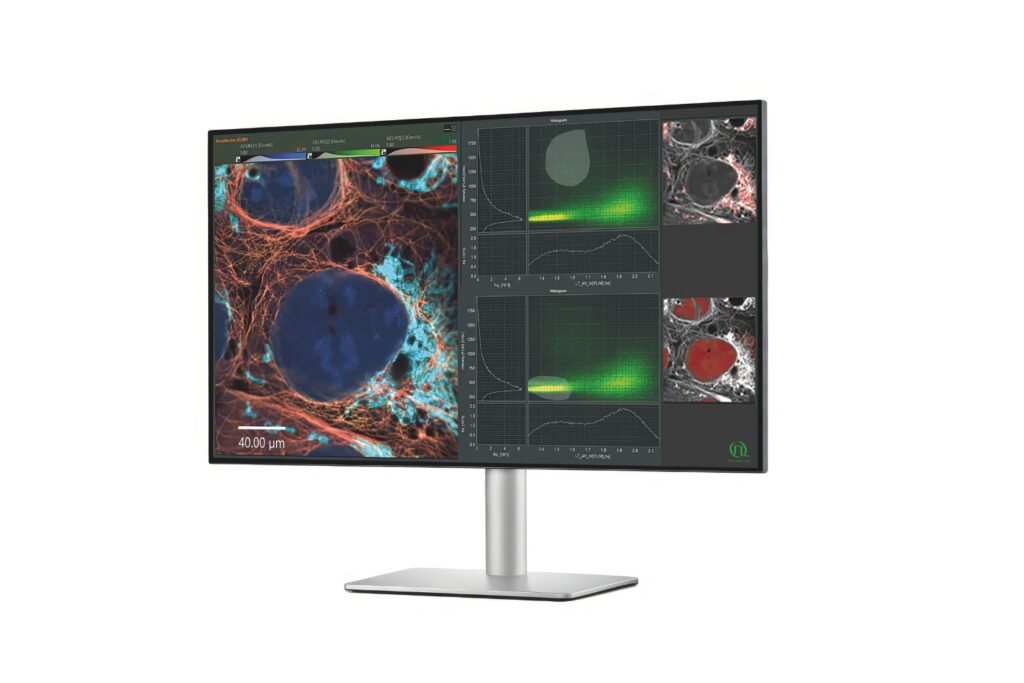
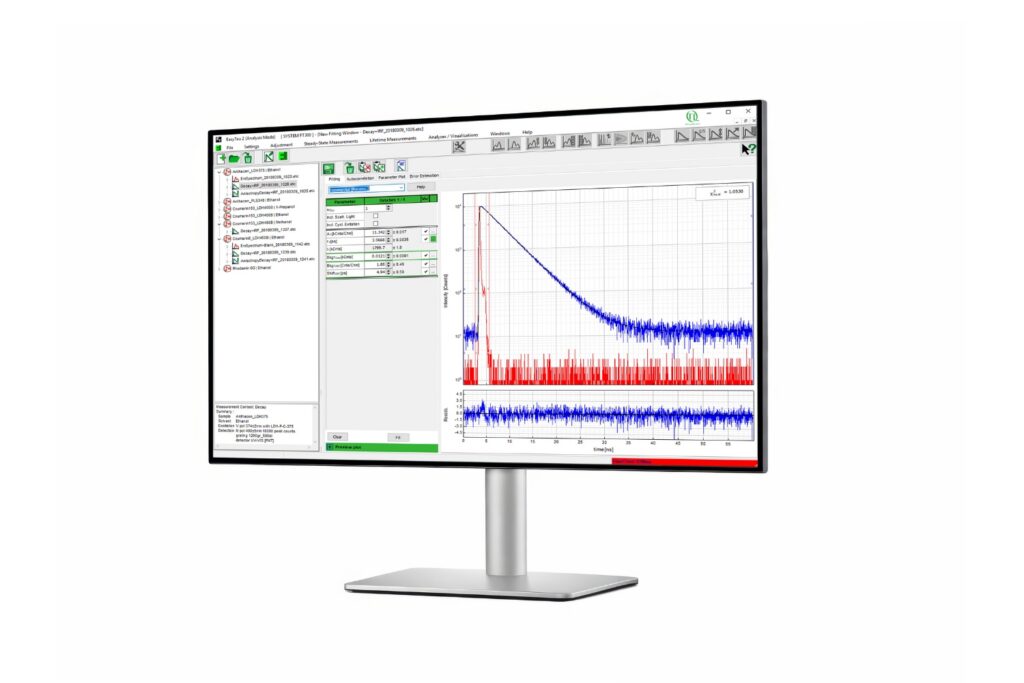
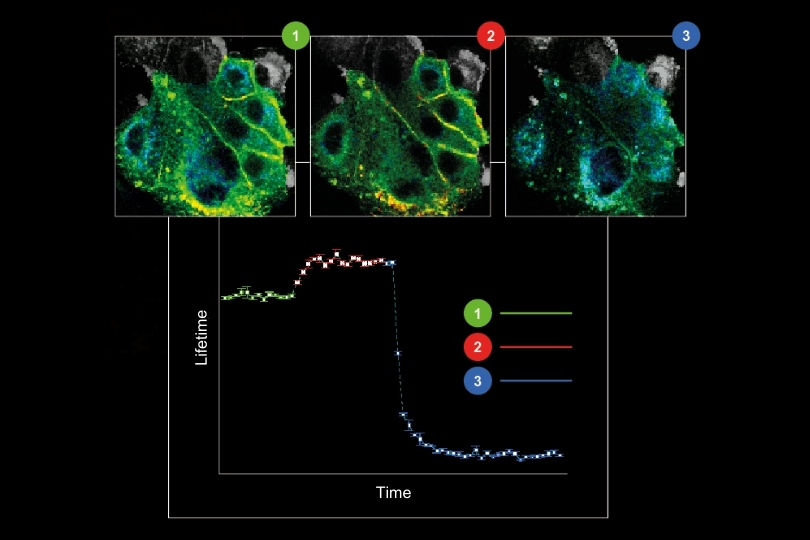
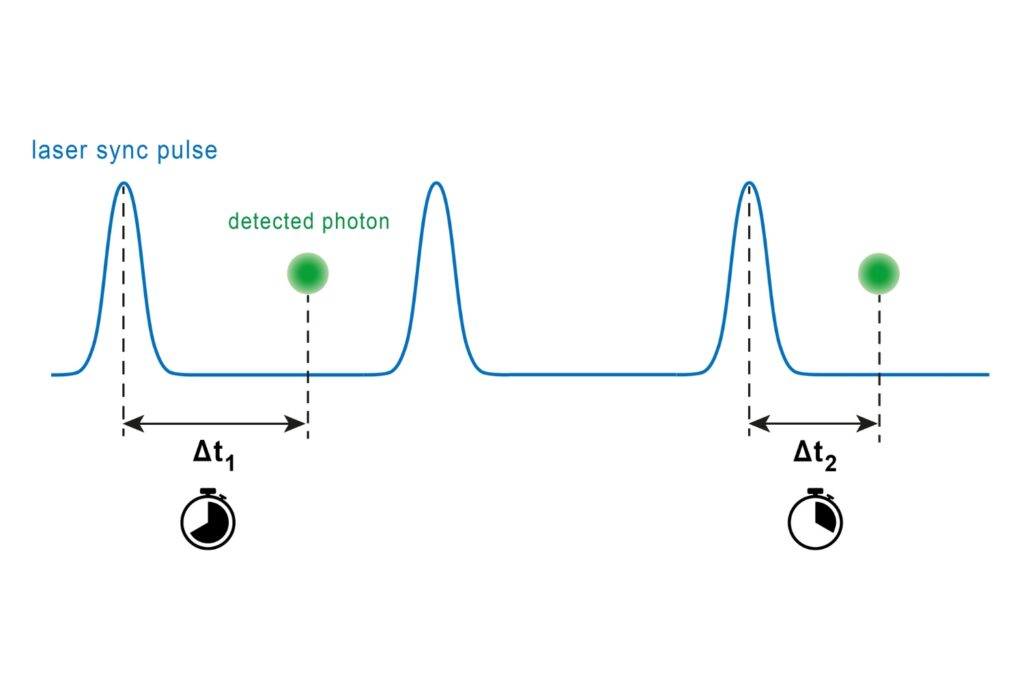
A fluorescence lifetime imaging microscope extends confocal microscopy into the time domain by recording fluorescence decay profiles using time-correlated single photon counting (TCSPC). Instead of relying solely on intensity, quantitative FLIM reconstructs the temporal distribution of detected photons in each pixel. Fluorescence lifetime provides a parameter that is largely independent of fluorophore concentration and excitation intensity. This enables quantitative measurements of molecular interactions and environmental changes. In FLIM-FRET experiments, the shortening of the donor lifetime directly reflects energy transfer efficiency. In single-molecule FRET, even small distortions of the decay curve can bias calculated efficiencies.
The accuracy of fluorescence lifetime measurements therefore depends on precise photon timing and faithful decay reconstruction. Count rate handling and timing integrity ultimately determine whether quantitative FLIM remains reliable under realistic imaging conditions.
System-Level Requirements for High Count Rate FLIM
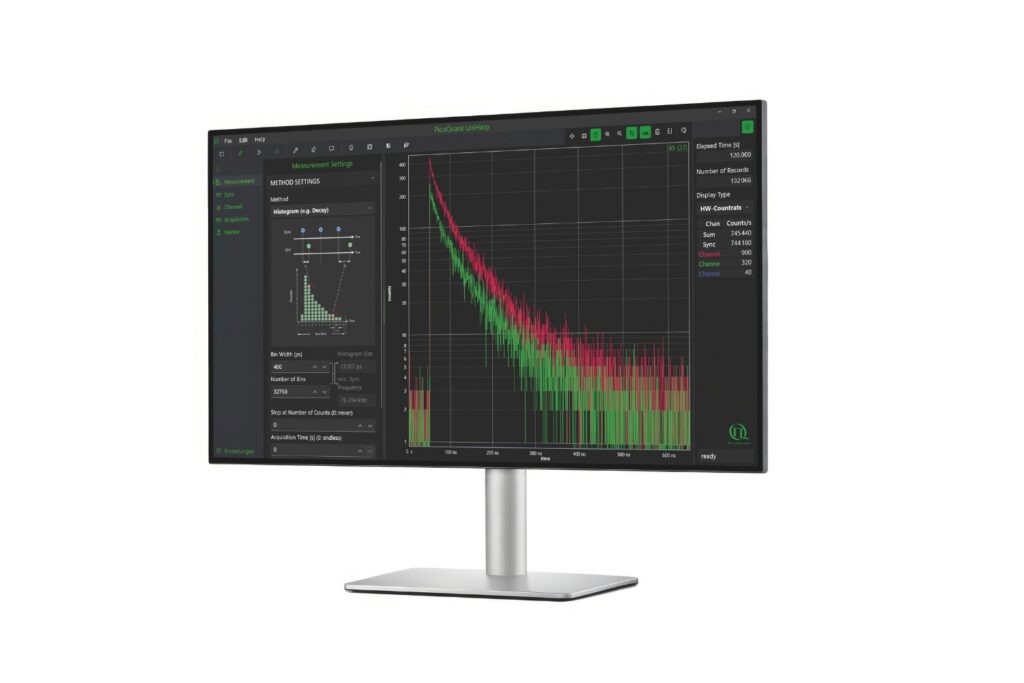
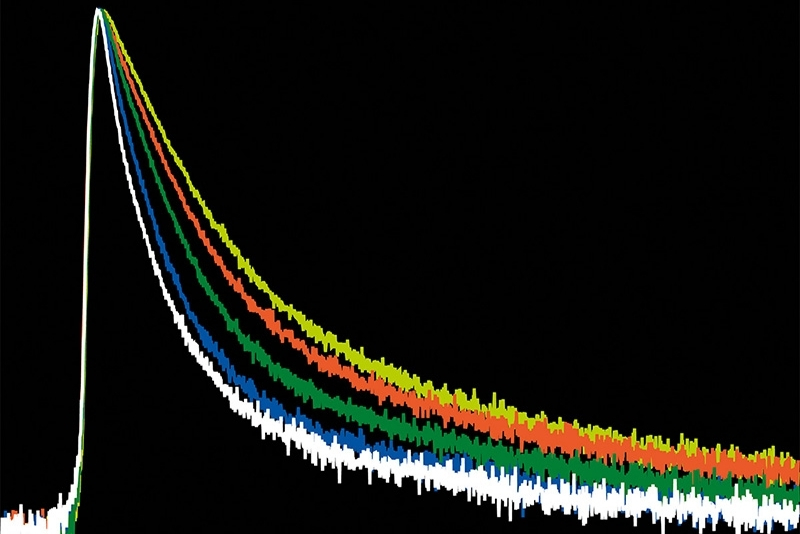
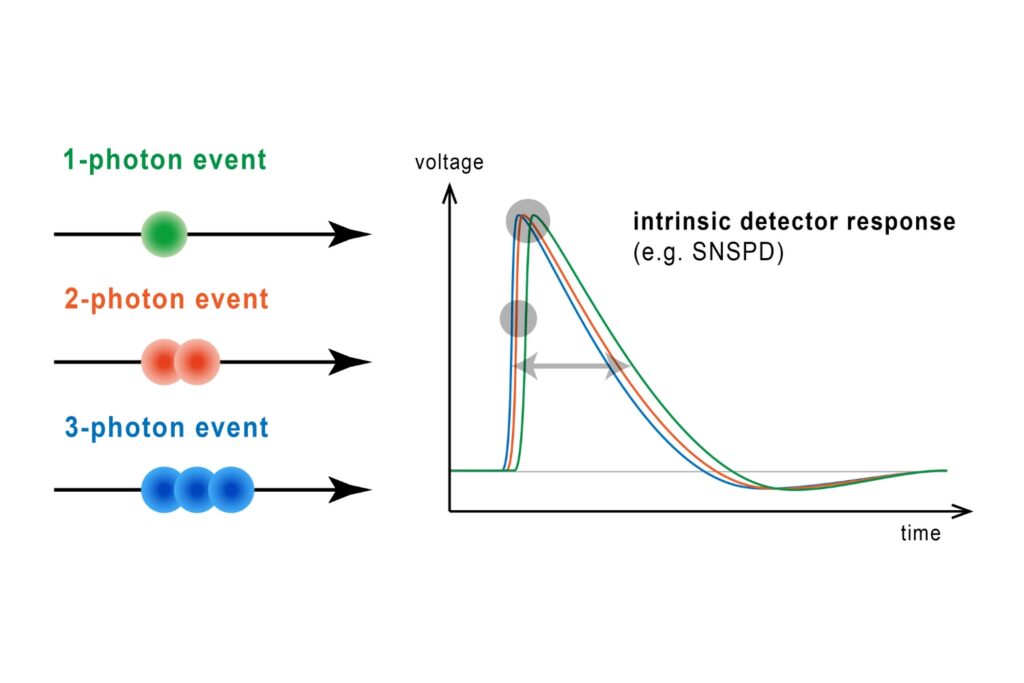
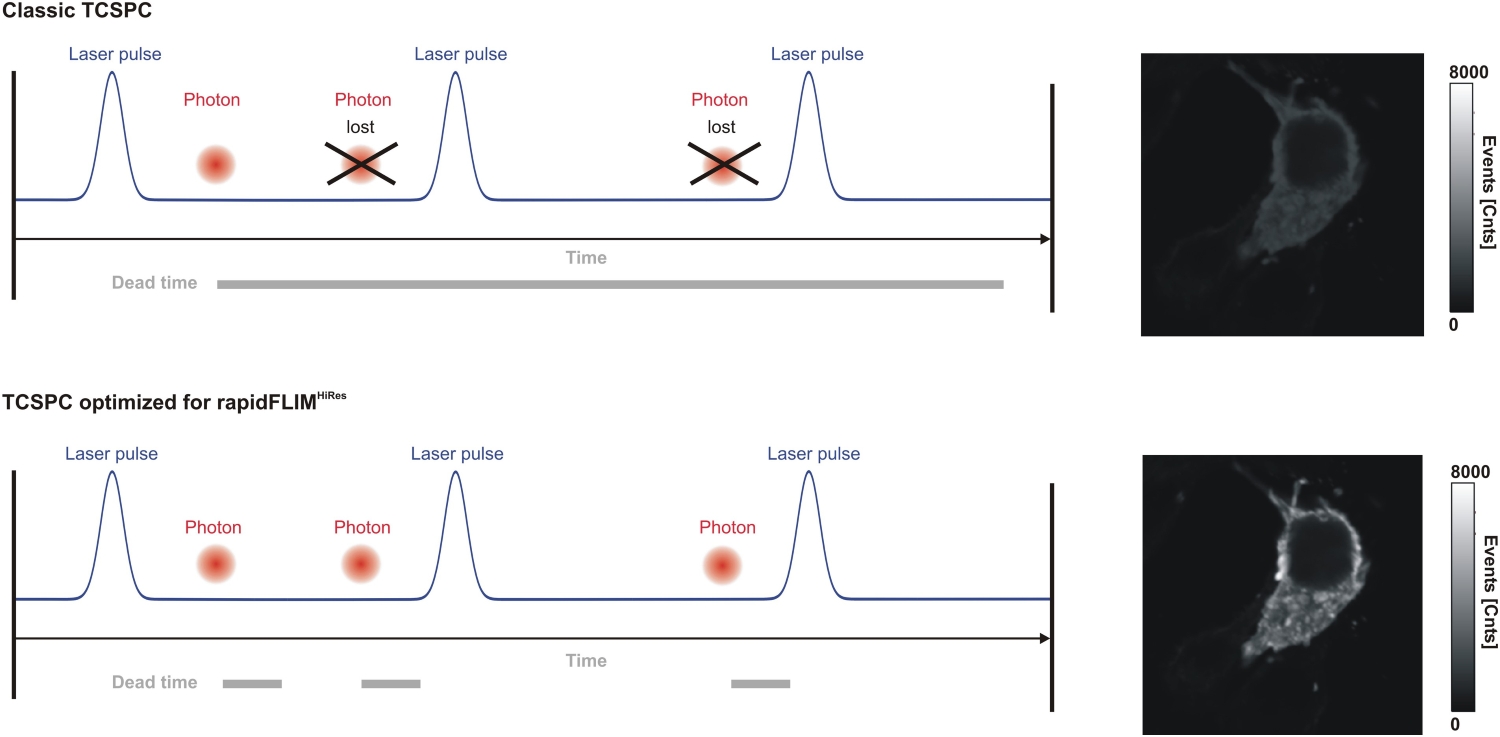
Lifetime precision improves with the number of detected photons, which makes high count rate FLIM attractive for rapid imaging and low-noise decay reconstruction. At the same time, increasing photon flux places fundamental demands on detector and timing electronics. Each detected photon must be registered and temporally assigned relative to the excitation pulse. If the detection system cannot process incoming photons fast enough, systematic distortions occur. Dead time effects and photon pile-up lead to preferential detection of early photons in the decay, artificially shortening measured lifetimes.
High count rate performance therefore requires more than sensitivity. A time-resolved confocal microscope must sustain elevated photon rates without compromising temporal fidelity. The relevant parameter is not peak brightness alone, but the ability of the system to maintain accurate decay reconstruction under continuous, realistic imaging conditions.
Preserving Lifetime Accuracy Under Real Imaging Conditions
In practical imaging scenarios, photon flux is rarely constant or moderate. Bright structures, dense labeling, or rapid acquisition can push detection rates into regimes where timing electronics become limiting. Under such conditions, systematic errors outweigh random noise. As count rates approach the processing limits of a TCSPC system, detector dead time results in preferential registration of early photons. This photon pile-up alters the reconstructed decay profile and artificially shortens the apparent lifetime, although the underlying molecular dynamics remain unchanged.
These effects are particularly critical in quantitative applications. In FLIM-FRET experiments, donor lifetime shortening is used to calculate energy transfer efficiency. In single-molecule FRET, the extracted lifetime directly influences distance estimations and interaction models. Even small systematic deviations in decay reconstruction can therefore propagate into significant errors in biological interpretation.
Preserving lifetime accuracy under realistic imaging conditions requires a system that sustains high photon count rates while maintaining temporal integrity. Sensitivity alone does not guarantee quantitative reliability. The combination of efficient photon collection, short detector dead time, and stable timing electronics ultimately determines whether high count rate FLIM remains quantitatively reliable.
A Single Photon Counting Confocal Microscope Designed for High Count Rate FLIM
If high count rate FLIM requires sustained photon handling, minimal dead time, and stable timing performance, these constraints must be addressed at the architectural level of the microscope. Luminosa was developed as a single photon counting confocal microscope specifically engineered for time-resolved applications.
Rather than treating sensitivity and timing as separate components, the system integrates detector technology, TCSPC electronics, and optical design into a coherent architecture. This integration becomes critical when fluorescence lifetime measurements are performed under elevated photon flux, where detector recovery time and timing precision directly influence decay reconstruction.

Detector and Timing Architecture for High Count Rate FLIM
Luminosa incorporates the MultiHarp High-Throughput Multichannel Time Tagging & TCSPC Unit with an ultra-short dead time of 650 ps. This short recovery time enables sustained count rates of up to approximately 78 Mcps and supports rapidFLIMHiRes acquisition at high frame rates.
The relevance of these specifications lies not in peak values alone, but in the ability to maintain decay fidelity as photon flux increases. Short detector dead time reduces preferential registration of early photons and supports accurate lifetime reconstruction across a broad dynamic range. For high count rate FLIM, sustained timing stability is a prerequisite for quantitative fluorescence lifetime imaging rather than a secondary performance parameter.
Optical Efficiency and Detected Photon Yield in Confocal FLIM
High count rate handling must be complemented by efficient photon collection. The optical layout is optimized to minimize optical elements and maximize detection efficiency. Luminosa allows switching between galvo scanning and piezo objective scanning. By bypassing scanning mirrors, piezo operation can increase detected photon yield by approximately 20 to 30 percent, depending on wavelength. Increased photon yield directly improves lifetime precision without requiring higher excitation power, thereby supporting accurate decay reconstruction under demanding imaging conditions.
Implications for Quantitative Fluorescence Lifetime Imaging and smFRET
High count rate capability defines the quantitative operating regime of a FLIM microscope. When photon flux increases, lifetime accuracy depends on sustained timing stability, short detector dead time, and efficient photon detection. Without coherent system design, decay distortion limits the interpretability of time-resolved data. For quantitative fluorescence lifetime imaging and smFRET, preserving decay fidelity under realistic imaging conditions is therefore not a performance detail. It is a prerequisite for reliable parameter extraction.