Key Highlights
- High-rate FLIM enabled full-frame sodium imaging at 0.5 Hz in acute hippocampal brain slices.
- Short-dead-time TCSPC electronics and hybrid photon counting detectors minimized lifetime distortions at elevated count rates.
- Fluorescence lifetime measurements remained stable under ischemia-induced cell swelling, while intensity-based readouts became unreliable.
- The modular detection and timing architecture demonstrates how existing laser scanning microscopes can be upgraded for quantitative, intensity-independent FLIM.
Why Intensity-Based Imaging Fails Under Cellular Stress
Quantifying intracellular sodium in living tissue is technically demanding. Available Na⁺ indicators emit comparatively few photons, and classical TCSPC-based FLIM often requires long integration times to obtain stable decay fits. Under dynamic biological conditions, this quickly becomes limiting.
The deeper problem emerges under stress. During metabolic inhibition or ischemia, neurons swell. Changes in cell volume alter dye concentration and therefore fluorescence intensity. What appears as a change in ion concentration may instead reflect volumetric or optical artifacts.
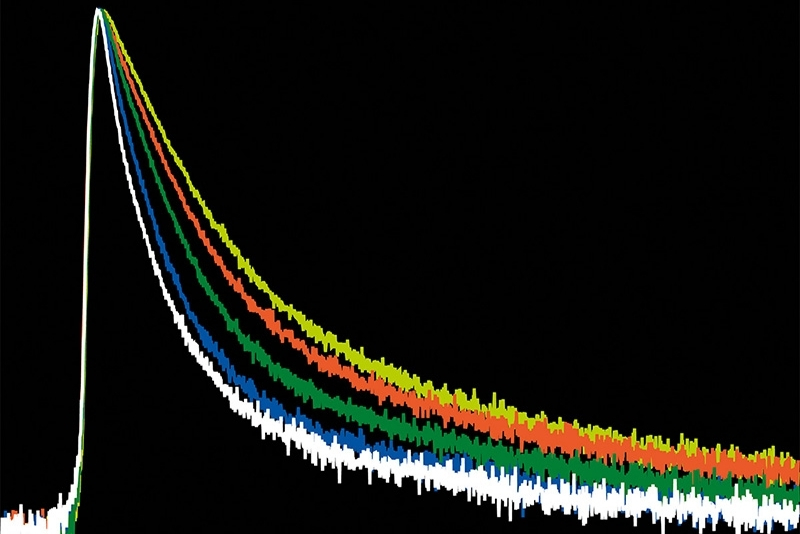
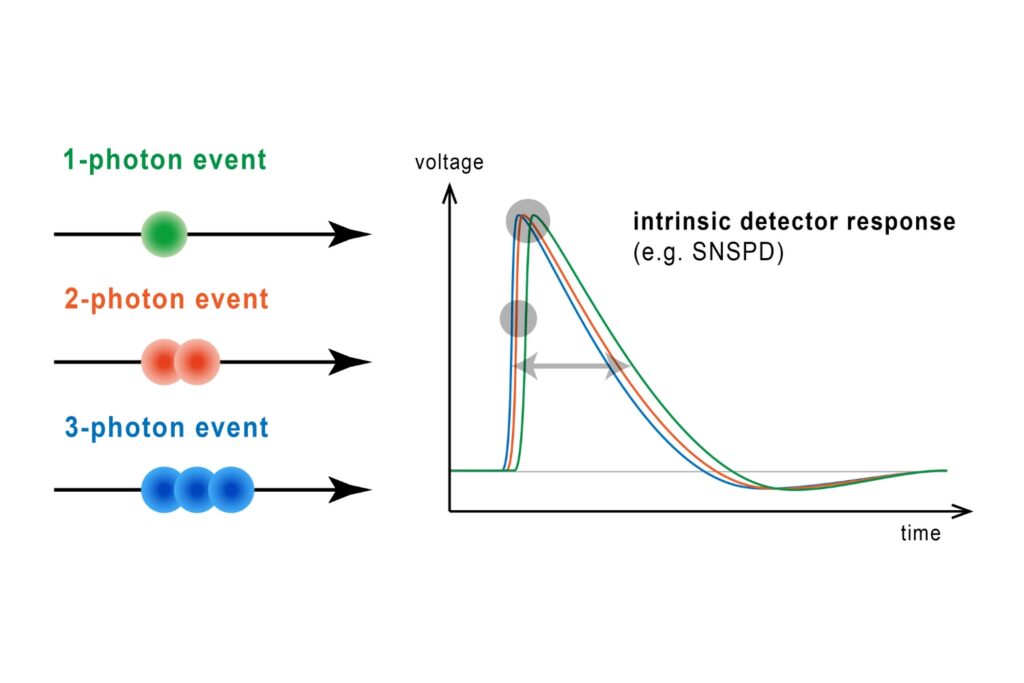
In addition, conventional TCSPC electronics impose count-rate limits. When photon rates approach a few percent of the laser repetition rate, detector dead time and pulse pile-up begin to distort decay histograms. Apparent lifetimes shorten artificially, particularly with dim probes that require stronger excitation.
Meyer et al., Journal of Neuroscience (2022), addressed precisely this challenge: how to perform dynamic, quantitative Na⁺ imaging in acute hippocampal slices without intensity-induced bias. The biological question was important. The technical solution was decisive.
Upgrading Detection and TCSPC in an Existing Confocal
The key insight was that the limitation was not the microscope optics. It was the detection and timing architecture.
Two technical aspects are decisive when pushing FLIM into higher count rate regimes with dim probes:
- Extremely short electronic dead time
- Clean detector pulse response with minimal afterpulsing
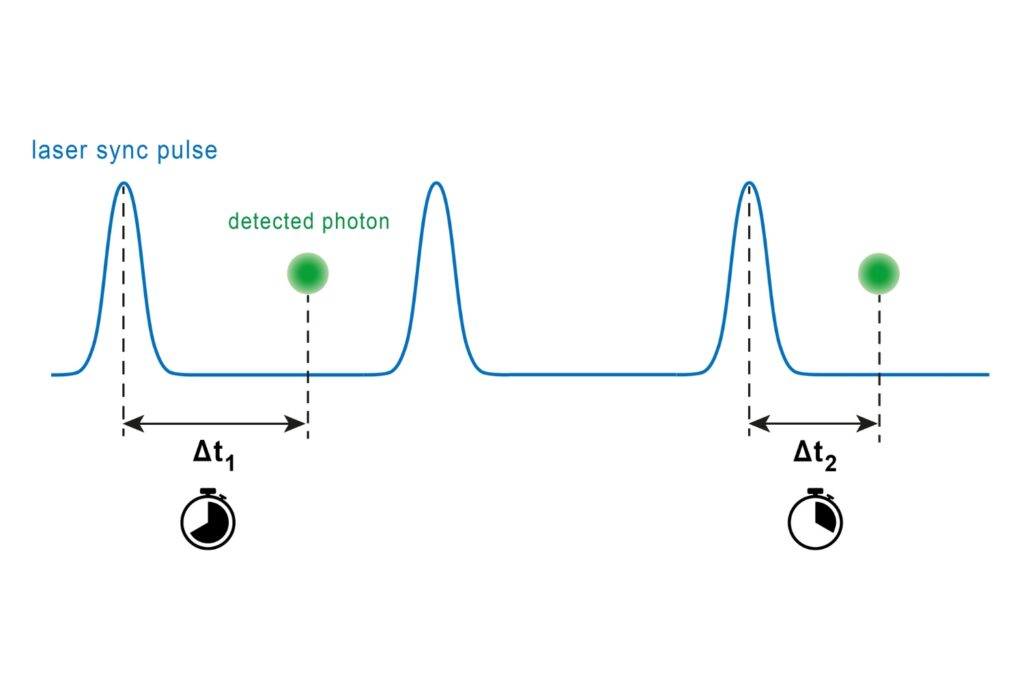
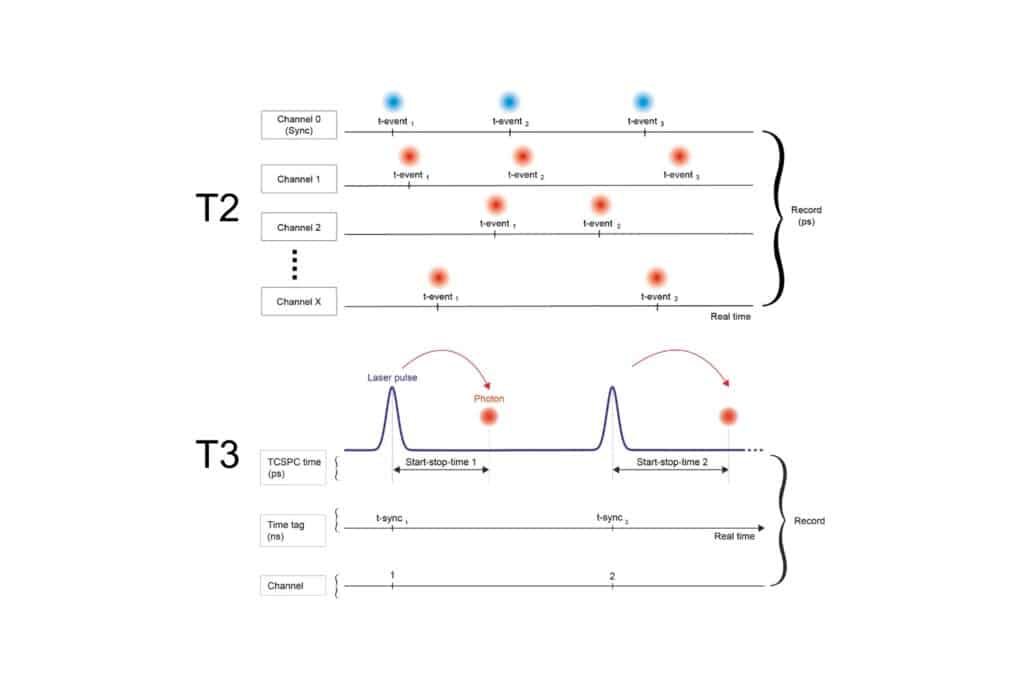
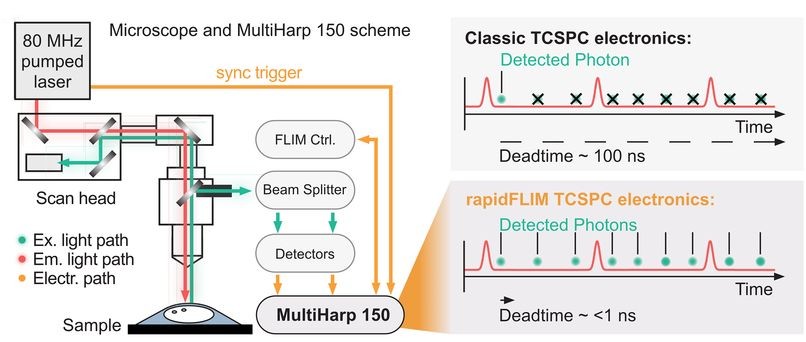
By combining hybrid photomultiplier detectors with rapid time tagging & TCSPC electronics featuring sub-nanosecond dead time, the system allowed photon count rates that would saturate classical architectures. In this configuration, dead-time artifacts were largely eliminated, and electrical pulse pile-up could be corrected directly within the fitting model. All photons were recorded in a Time-Tagged Time-Resolved format, preserving full temporal information and enabling robust decay analysis. An improved model-based correction accounted for pulse merging effects directly within the fitting routine.
This architecture corresponds to the modular detection and timing configuration implemented in PicoQuant’s LSM Upgrade Kit. Instead of replacing the microscope, the approach externally adds picosecond pulsed excitation, single-photon counting detection, and high-throughput TCSPC acquisition to an existing laser scanning microscope.
The result is not a new microscope. It is a new level of temporal precision attached to an existing platform.
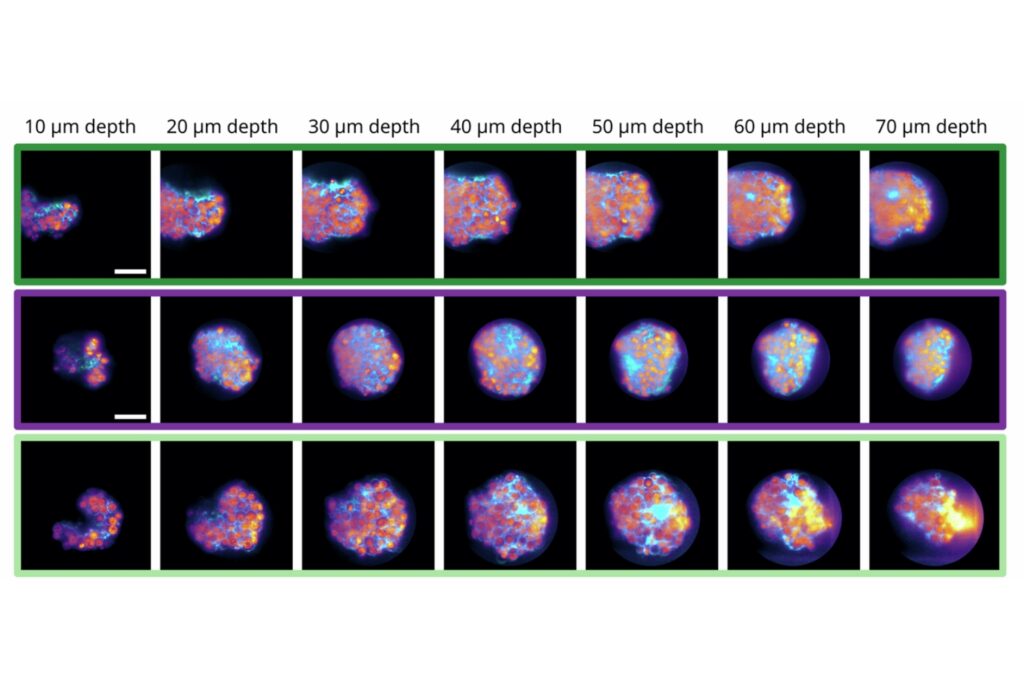
High-Rate Sodium FLIM in Living Brain Tissue
The upgraded configuration was validated under demanding biological conditions.
Using the sodium indicator ING2, the authors performed lifetime calibration in vitro and in situ. The fluorescence lifetime increased with intracellular Na⁺ concentration and remained largely insensitive to pH, potassium, calcium, and viscosity changes within the tested range.
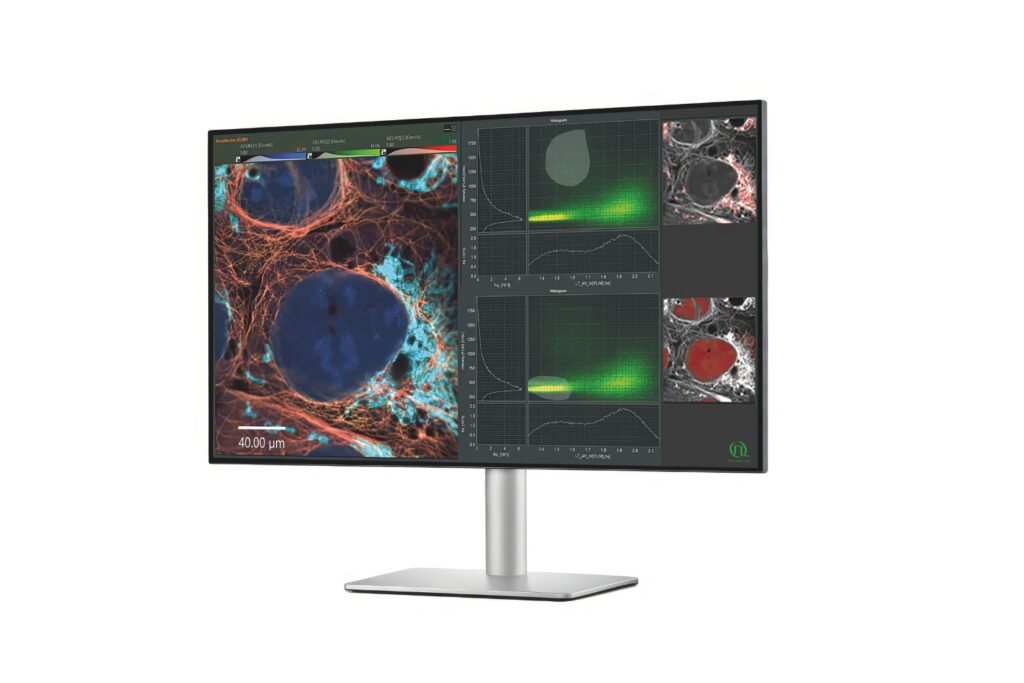
In acute hippocampal slices, the system enabled full-frame FLIM at 0.5 Hz and reduced-frame acquisition at 2 Hz, despite the low brightness of the probe. These acquisition rates exceed typical Na⁺ FLIM performance with classical TCSPC setups.
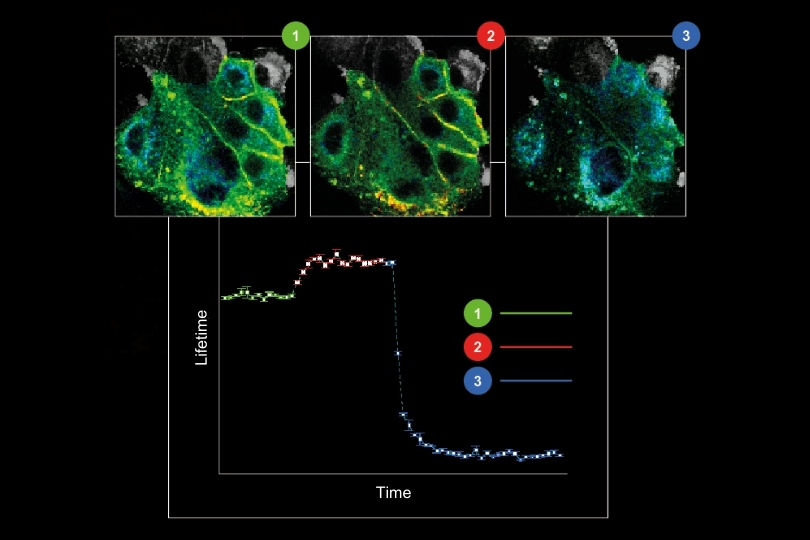
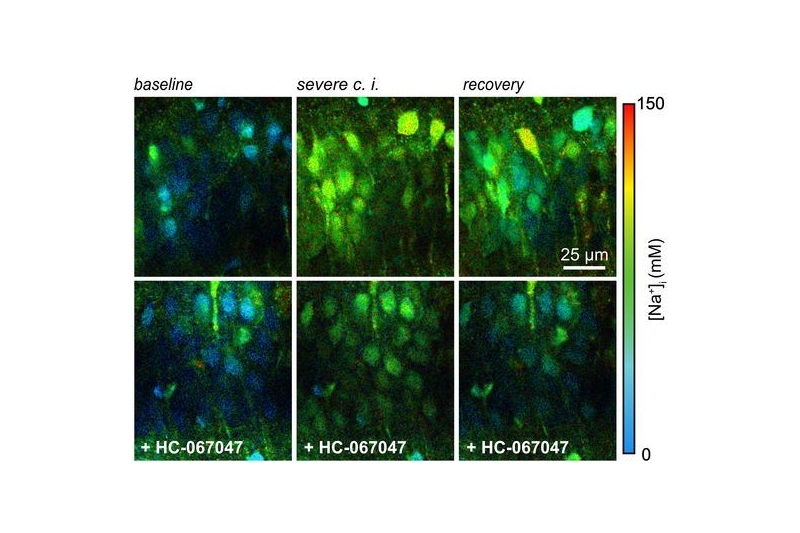
Transient chemical ischemia was then induced by inhibiting mitochondrial respiration and glycolysis. Intracellular Na⁺ rose dramatically, scaling with the severity of metabolic inhibition.
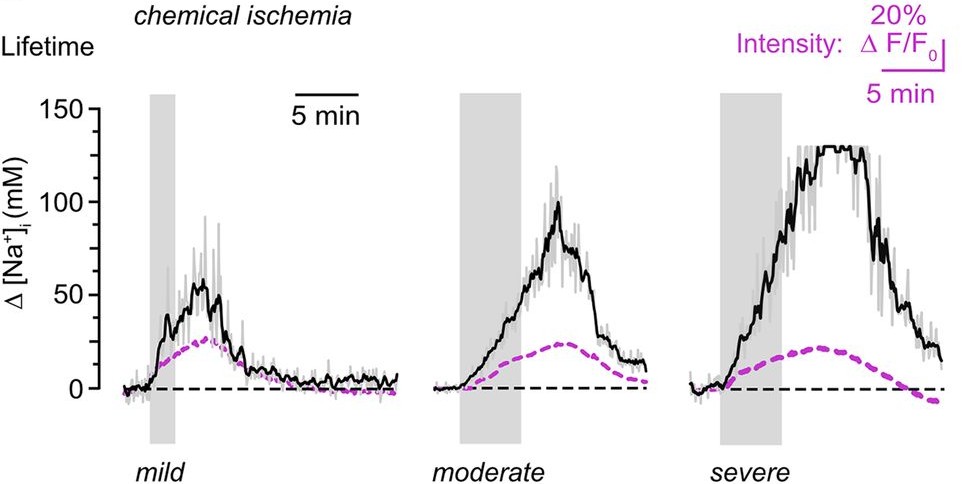
Here, the difference between intensity and lifetime became evident. While lifetime analysis reported progressively larger Na⁺ increases, fluorescence intensity did not scale accordingly. As cell swelling progressed, intensity changes no longer reflected true ion concentrations.
Lifetime measurements remained stable and intensity-independent. This robustness allowed the authors to identify a substantial contribution of TRPV4 channels to ischemia-induced Na⁺ loading. Inhibition of TRPV4 reduced both neuronal swelling and sodium influx.
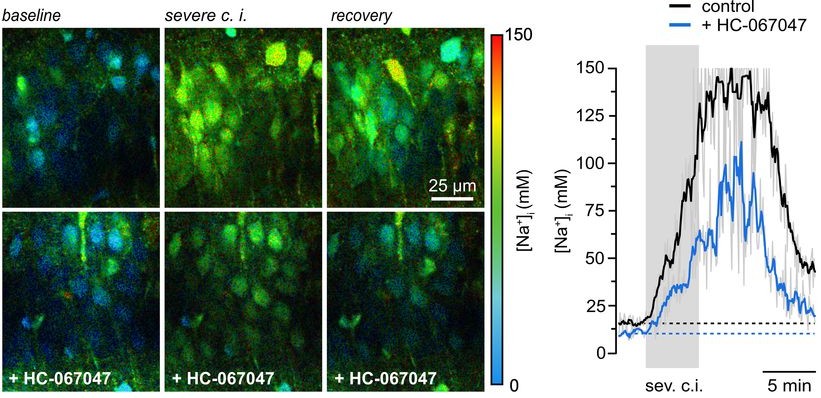
The biological insight was significant. Equally important was the demonstration that quantitative, high-rate Na⁺ FLIM could be achieved on an existing confocal microscope when detection and TCSPC performance were upgraded appropriately.
Why This Matters for Existing LSM Platforms
Many laboratories operate advanced laser scanning microscopes that lack robust time-resolved capabilities. Replacing these systems to enable quantitative FLIM or FCS is often unnecessary.
This study shows that the limitation lies not in the optics, but in detector dead time and TCSPC performance. By upgrading excitation, detection, and timing, an existing confocal can be transformed into a quantitative lifetime imaging platform.
Extending your microscope does not require replacing it. It requires improving how photons are detected and timed.
Instrumentation Used in This Study by PicoQuant
The experiments were performed using a configuration of the LSM Upgrade Kit, which adds time-resolved fluorescence capabilities to existing laser scanning microscopes. The setup included the following core components:
- MultiHarp 150 multichannel time-tagging and TCSPC unit providing picosecond timing precision and ~650 ps dead time for high-count-rate lifetime acquisition
- PMA Hybrid 40mod single-photon counting detector enabling clean pulse response and stable timing at elevated photon flux
- Picosecond pulsed diode laser at 640 nm delivering synchronized excitation matched to the sodium indicator
- SymPhoTime 64 software for photon acquisition and quantitative fluorescence lifetime analysis

Together, this configuration represents the detection and timing architecture required for quantitative TCSPC-based FLIM on a confocal laser scanning microscope.
Learn how the LSM Upgrade Kit adds quantitative FLIM and FCS capabilities to your existing laser scanning microscope.