Photon Counting Detectors


Complete confocal fluorescence microscope that empowers researchers to advance quantitative functional imaging from individual molecules to cells and tissues.

Modular, customizable, time-resolved confocal microscope with single-molecule sensitivity for life and materials science.

Compact FLIM and FCS upgrade kit that adds advanced functional imaging and correlation analysis to existing laser scanning microscopes.

Designed for flexible, sensitive, and precise steady-state and time-resolved spectroscopy across the UV to NIR range and time scales from picoseconds to milliseconds.

Modular lifetime spectrometer designed for flexible fluorescence and photoluminescence measurements in both materials and life science research.

Add spectral and time-resolved photoluminescence to your setup through flexible microscope–spectrometer coupling options.

Get the most out of superconducting nanowire detectors in large-scale quantum communication and computing experiments requiring precise multichannel timing.

Boost your time-resolved experiments with a flexible, high-precision time tagging and TCSPC unit for materials science and quantum sensing.

Scale your photonic quantum computing and detector characterization setups while maintaining performance, flexibility, and high data throughput.

Compact 3-color picosecond laser delivering flexible ns to ms excitation with cost-effective multicolor performance and straightforward operation.

Smart picosecond laser diode heads covering UV-A to NIR, providing the right combination of power, pulse width, and diode type for any time-resolved technique.

VisUV provides clean short pulses and stable timing across key UV and visible wavelengths, including deep UV lines as well as 488 nm and 532 nm.

Enhance your single-photon counting experiments with wide dynamic range and excellent timing precision in the UV and visible even at the highest count rates.

Capture even the weakest signals over large areas with maximum dynamic range and enhanced low-light sensitivity in a compact detector design.

Unlock spatially resolved single-photon detection with a 23-pixel SPAD array, combining low dark counts and precise time tagging for advanced experiments.
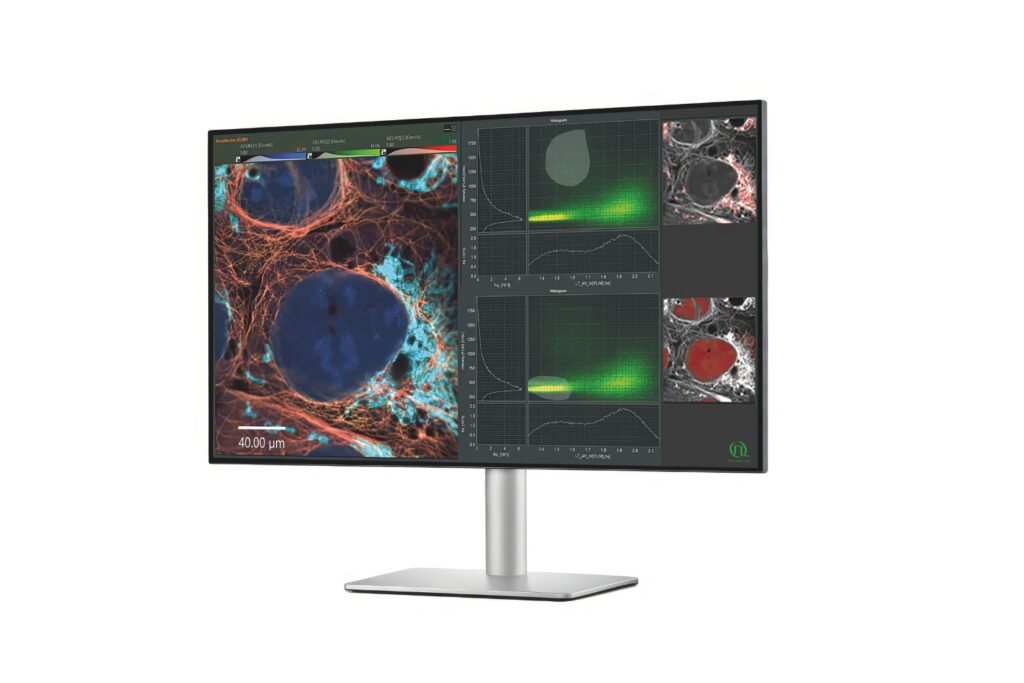
Advanced FLIM analysis software for fast, accurate interpretation of lifetime imaging data.
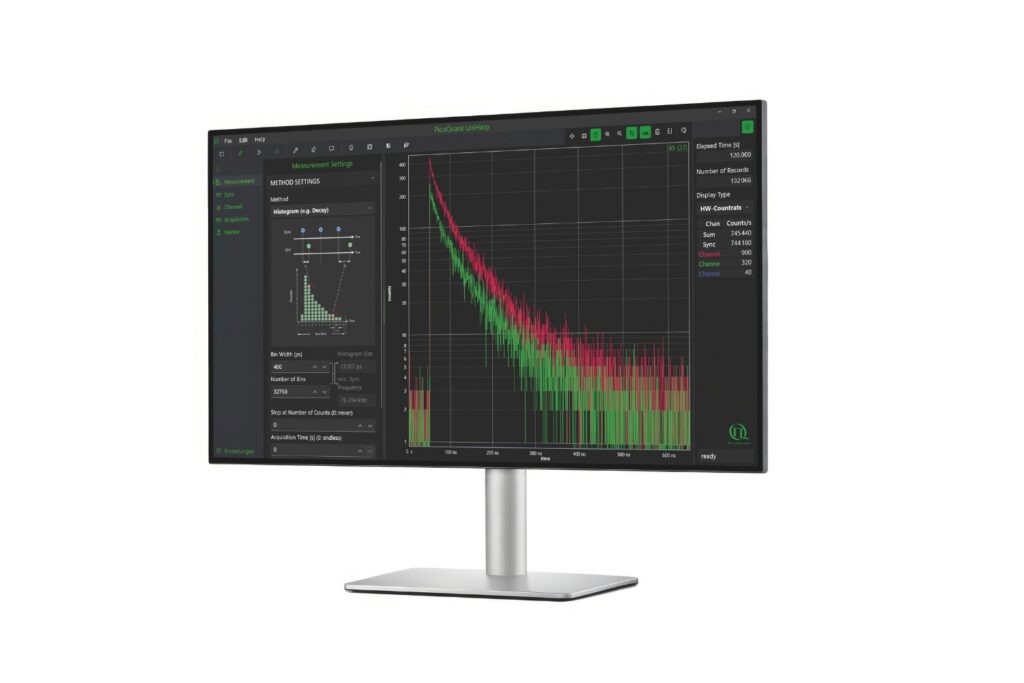
Intuitive, free software solution for real-time, high-precision photon data acquisition, visualization, and initial data analysis.
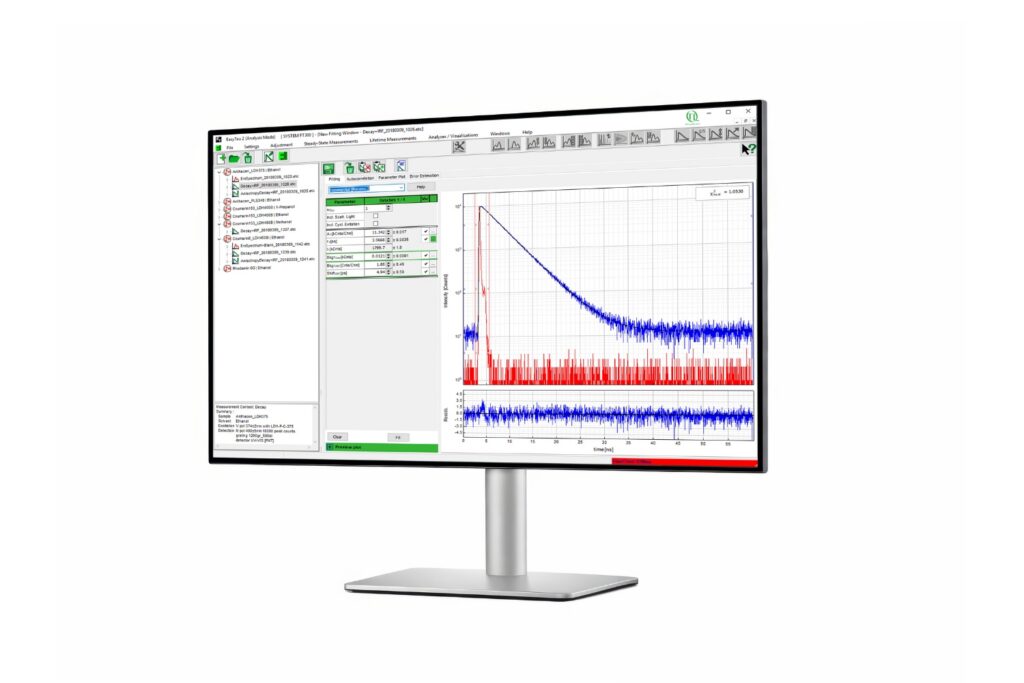
Advanced software for time-resolved fluorescence acquisition and analysis.

An imaging technique that uses fluorescence lifetimes to generate image contrast.
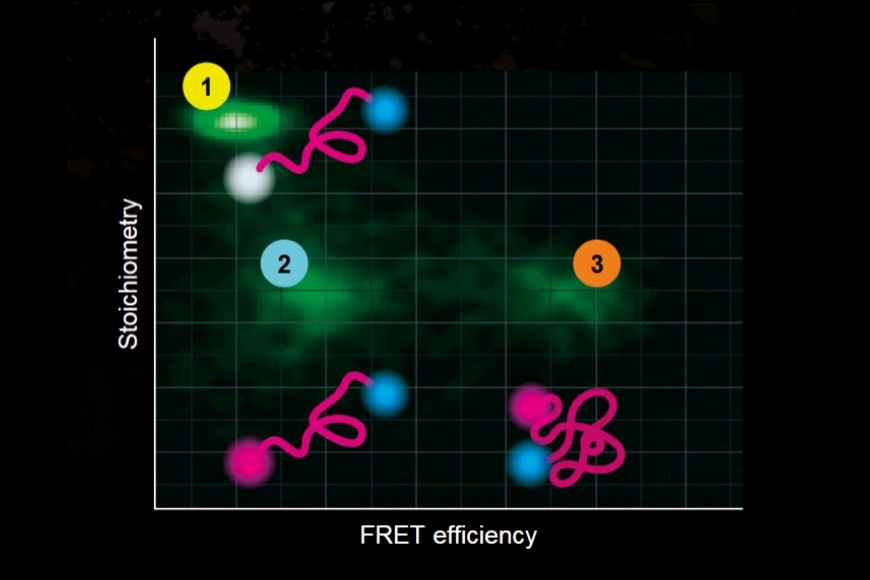
Investigating how proteins dynamically explore multiple conformational states that control biological function.

Investigating how biomolecules separate into dynamic liquid phases to organize cellular space and regulate biological function.
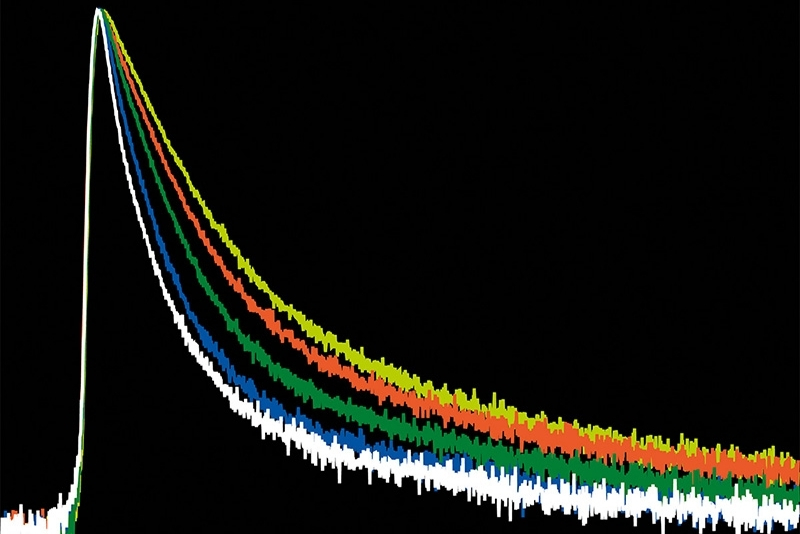
A time-resolved technique that measures photoluminescence lifetimes to reveal excited-state dynamics in materials.
Studying exciton dynamics, charge carrier processes, and structural properties through optical and time-resolved characterization methods.

Investigating charge-carrier lifetimes and recombination dynamics to enable precise optical characterization of material quality and device performance.
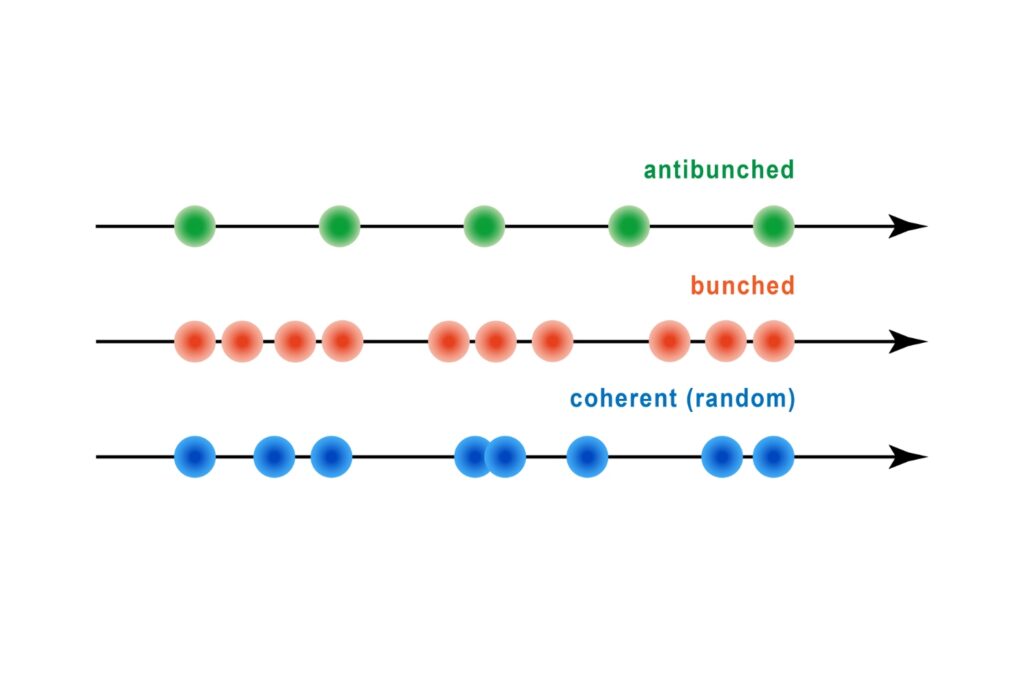
A quantum optical signature revealed by time-resolved photon correlation analysis to identify single-photon emission in materials and nanostructures.
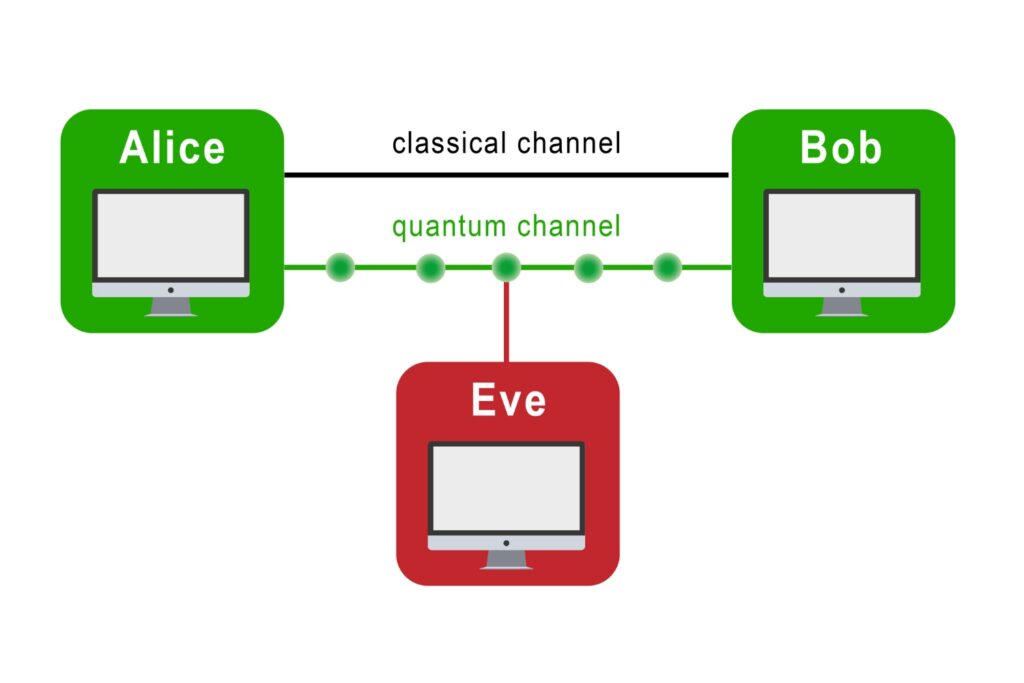
The transmission of information using individual photons, using quantum effects to ensure absolute security.
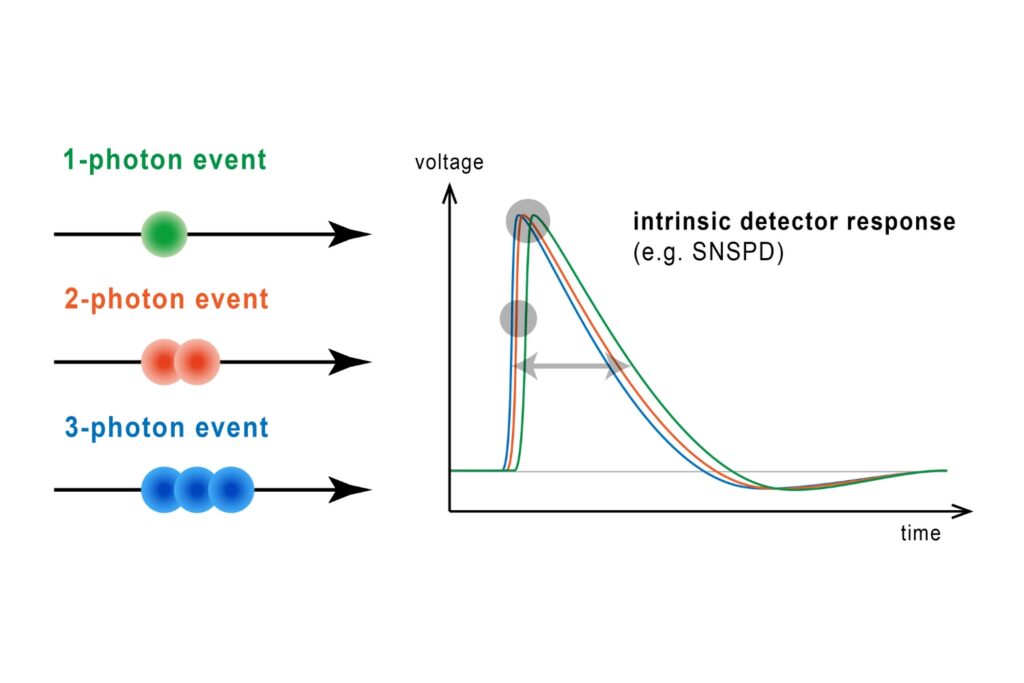
Quantifying photons per detection event enables direct access to photon-number statistics, providing insight into quantum and statistical properties of light.

An optical technique that analyzes light emission under electrical excitation to reveal electronic properties of electroluminescent materials.
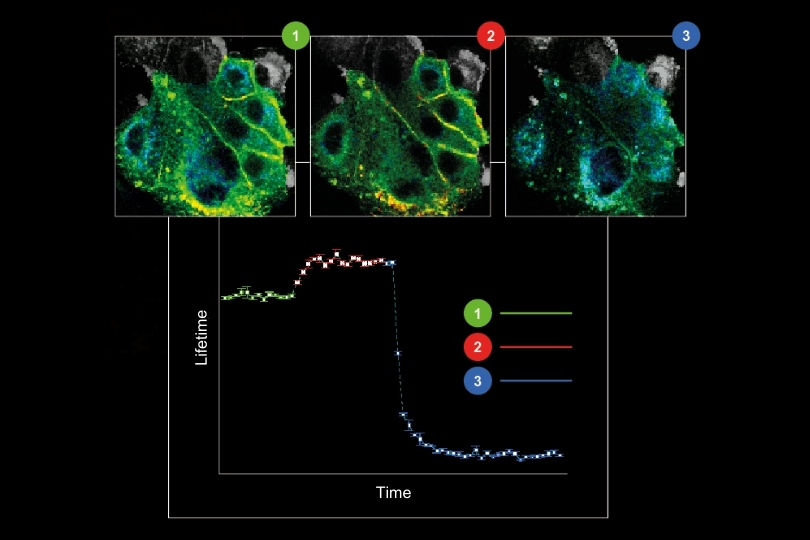
Monitoring environmental signals and trace compounds to understand dynamic changes in natural and engineered environments.
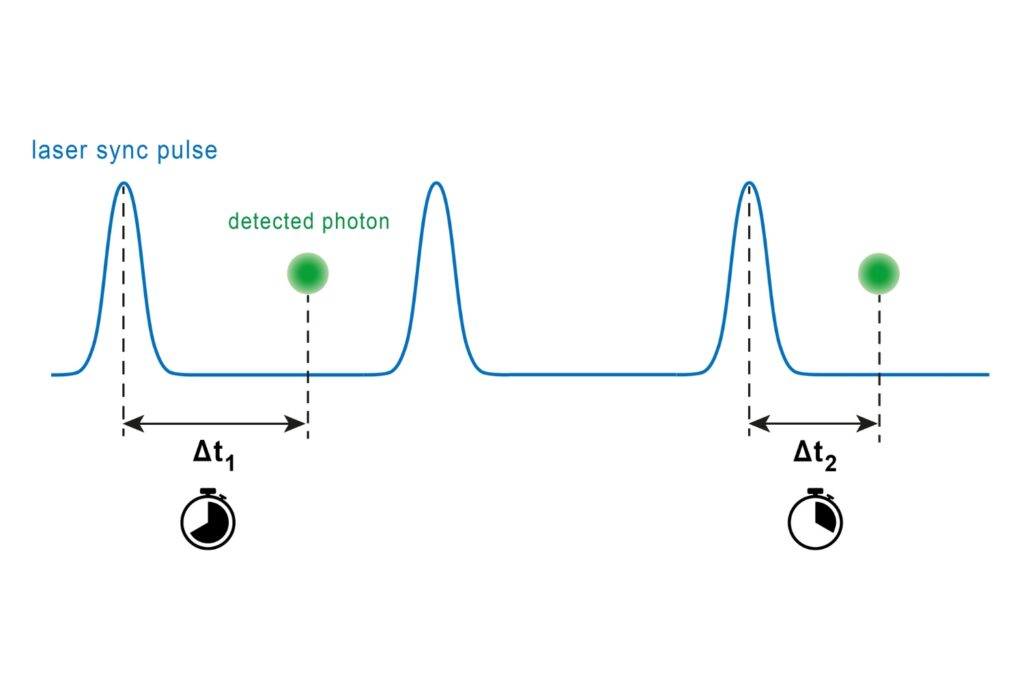
A photon timing technique that measures single-photon arrival times to resolve ultrafast dynamics in fluorescence, materials research, and quantum optics.
Fluorescence Recovery After Photobleaching (FRAP) is a fluorescence microscopy technique used to quantify the mobility and kinetic behavior of molecules within a sample, most commonly in living cells. It relies on the controlled photobleaching of fluorescently labeled molecules within a defined region, followed by observation of fluorescence recovery caused by the movement of unbleached molecules into that region.
This recovery process reflects molecular dynamics such as diffusion, transport, and binding interactions. Because FRAP directly measures how molecules redistribute over time, it provides quantitative insight into dynamic biological processes rather than static structural information.
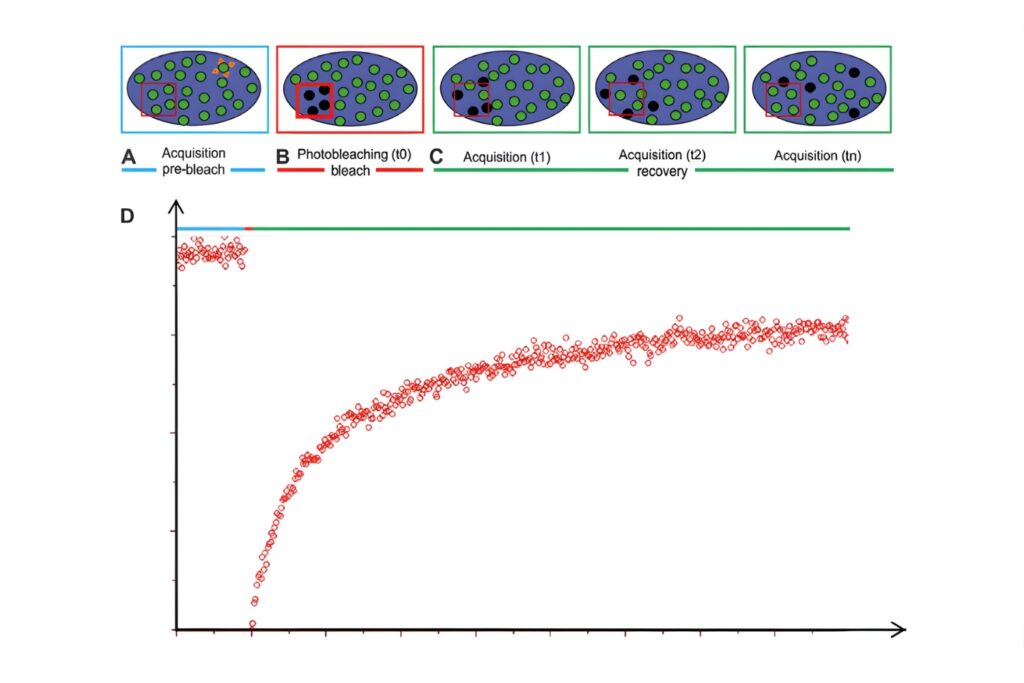
 Principle of FRAP experiments. A region of interest is photobleached using a high-intensity laser, and fluorescence recovery is monitored over time to analyze molecular mobility.
Principle of FRAP experiments. A region of interest is photobleached using a high-intensity laser, and fluorescence recovery is monitored over time to analyze molecular mobility.A FRAP experiment consists of four key steps:
Fluorescence images are recorded prior to bleaching to establish the initial intensity distribution and baseline signal within the region of interest (ROI), often across multiple frames to account for acquisition-related photobleaching.
A high-intensity laser pulse is used to selectively photobleach fluorophores in a defined ROI, irreversibly turning them non-fluorescent.
Fluorescence recovery is monitored over time as unbleached molecules redistribute into the bleached region through processes such as diffusion, active transport, or binding–unbinding dynamics.
The fluorescence recovery curve is analyzed to extract quantitative parameters such as diffusion coefficients, mobile and immobile fractions, and characteristic recovery times (e.g., t1/2), describing molecular mobility.
This process transforms photobleaching from a limitation into a powerful tool for studying molecular dynamics.
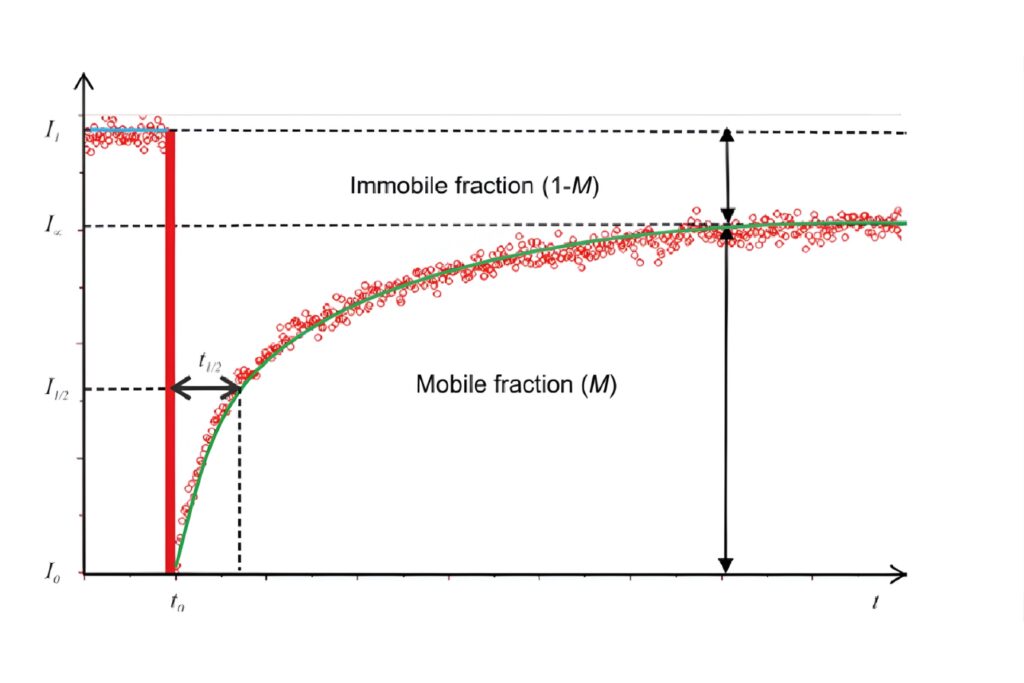 FRAP recovery curve illustrating half-time of recovery and separation of mobile and immobile molecular fractions.
FRAP recovery curve illustrating half-time of recovery and separation of mobile and immobile molecular fractions.The fluorescence recovery curve is central to FRAP analysis. It describes how fluorescence intensity changes over time within the bleached region. Key parameters include:
FRAP can be used to quantify and infer a wide range of molecular dynamics:
 Luminosa single photon counting confocal fluorescence microscope designed for quantitative time-resolved and single-molecule imaging.
Luminosa single photon counting confocal fluorescence microscope designed for quantitative time-resolved and single-molecule imaging.FRAP experiments require microscopy systems that enable precise control over excitation, photobleaching, and time-resolved image acquisition. Key requirements include high-intensity laser sources for controlled photobleaching, fast and stable imaging to capture recovery dynamics, and flexible region-of-interest (ROI) definition.
Modern confocal systems such as Luminosa integrate these requirements into a unified platform, enabling reliable and reproducible FRAP experiments within a controlled workflow, that guides users through photobleaching and recovery measurements while maintaining optimal imaging conditions. By combining precise laser control, flexible ROI definition, and automated acquisition routines, Luminosa supports the study of molecular mobility and dynamic processes directly within a confocal imaging environment. In addition, adaptive workflows and software-controlled acquisition allow users to adjust excitation power between imaging and photobleaching steps, helping to reduce unnecessary photodamage while preserving quantitative accuracy.
Please fill out the form below to request more information. You may also use it to inquire about pricing, availability, technical specifications, or discuss your specific application. Our sales team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information about our products and services. You may also use it to ask for pricing, availability, technical specifications, or any other details relevant to your inquiry. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information and prices about our product. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required