Key Highlights
- Chronic cadmium exposure disrupts autophagosome–lysosome fusion in prostate epithelial cells.
- NF-κB–dependent stabilization of PLAC8 promotes BCL-xL–mediated survival signaling during transformation.
- Quantitative confocal colocalization analysis reveals a progressive shift from LC3B–LAMP1 to LC3B–PLAC8 interactions.
- Pearson correlation–based imaging metrics provide mechanistic evidence for defective autophagy at the single-cell level.
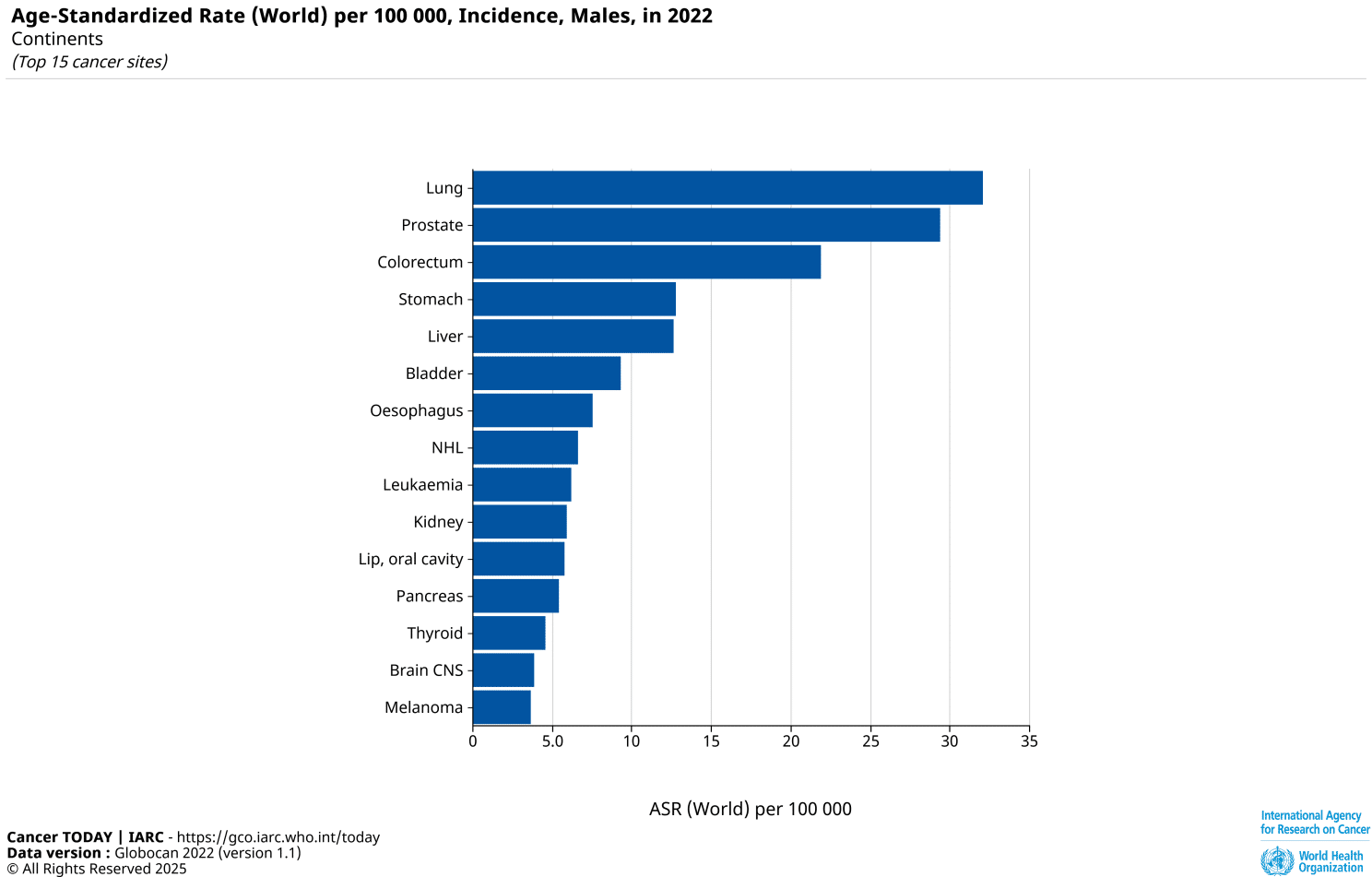
Prostate Cancer Among the Most Common Cancers Worldwide
According to GLOBOCAN 2022 data, prostate cancer ranks among the most frequently diagnosed malignancies in men worldwide, based on age-standardized incidence rates. Despite advances in screening and treatment, a detailed understanding of the molecular mechanisms driving tumor initiation and progression remains critical.
Prostate cancer development is shaped by both genetic predispositions and environmental exposures. Among the latter, cadmium (Cd) has repeatedly been associated with malignant transformation of prostate epithelial cells. Yet the molecular mechanisms linking chronic cadmium exposure to tumor-promoting survival pathways have remained incompletely understood.
In a recent study by Shukla et al., Sci. Adv. (2025), a regulatory signaling axis in cadmium-transformed prostate epithelial cells that connects NF-κB activation to impaired autophagy and apoptosis resistance was identified. Quantitative confocal colocalization analysis played an essential role in this discovery by enabling visualization of disrupted autophagosome–lysosome fusion at the single-cell level.
From Chronic Cadmium Exposure to Autophagy Dysfunction
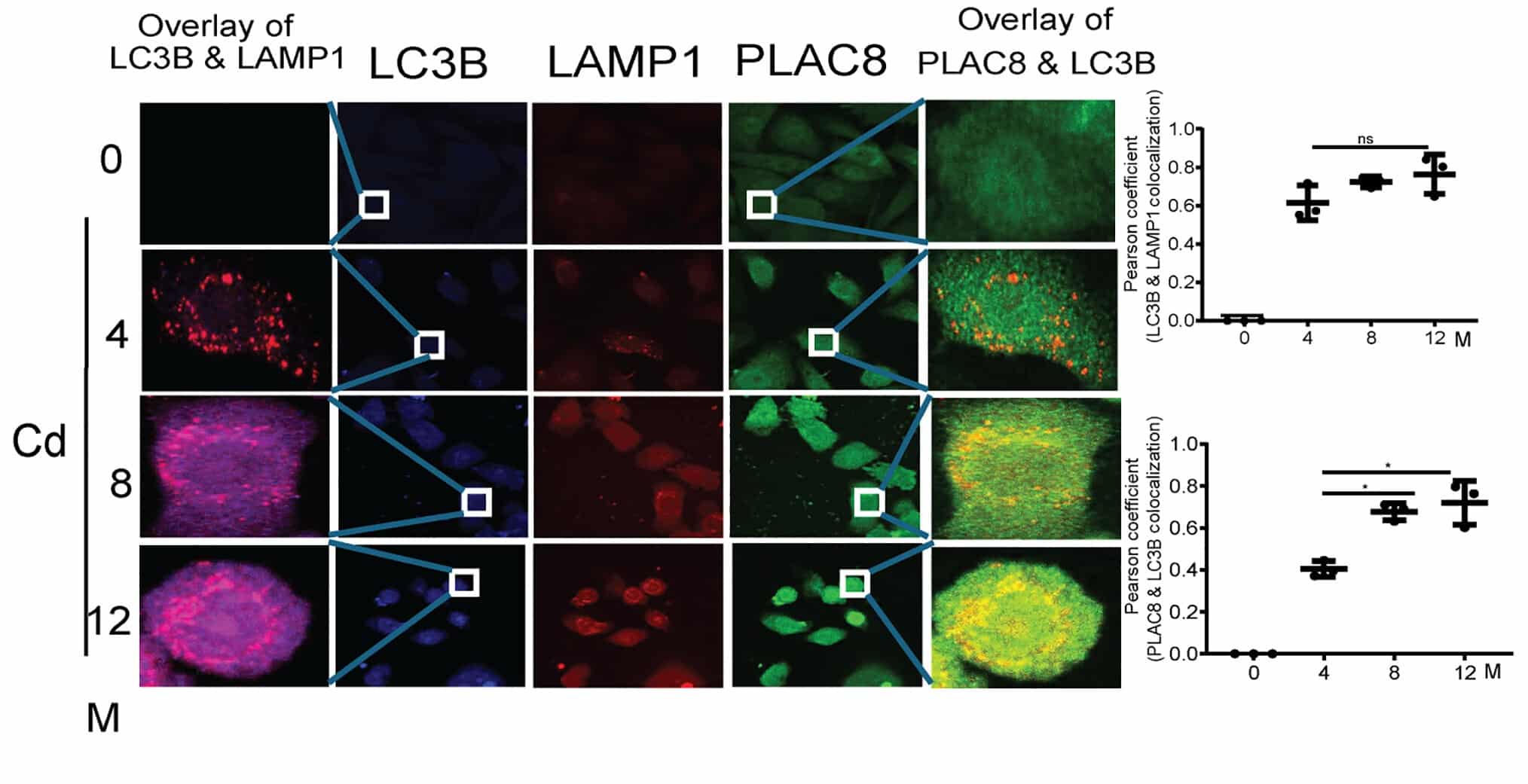
The authors used human RWPE-1 prostate epithelial cells chronically exposed to cadmium (10 μM) over 12 months, generating cadmium-transformed prostate epithelial (CTPE) cells. These transformed cells exhibited malignant characteristics and defective autophagic flux. Autophagy normally depends on the fusion of LC3B-positive autophagosomes with LAMP1-positive lysosomes, resulting in autolysosome formation and degradation of intracellular cargo. In CTPE cells, however, this process was disrupted.
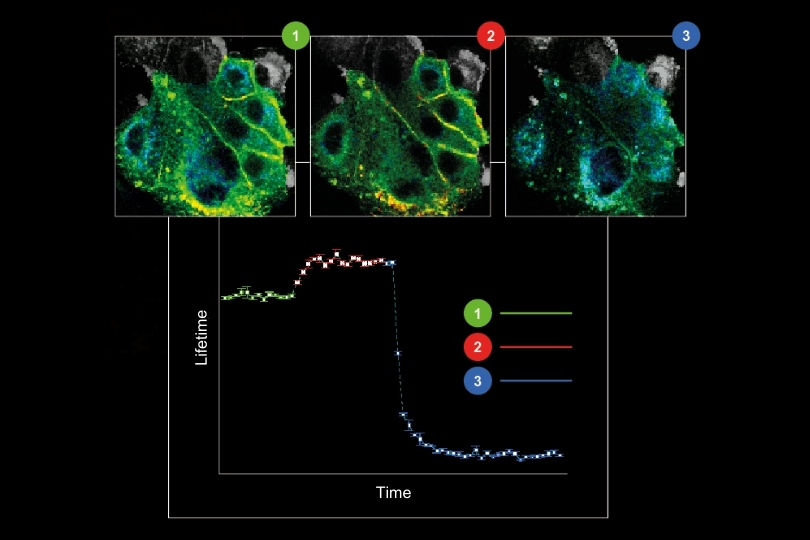
Instead of LC3B colocalizing with LAMP1, the study demonstrated increasing colocalization of LC3B with PLAC8 during progressive transformation stages. This shift impaired autolysosome formation and resulted in the accumulation of autophagosomes, indicating defective autophagy.
The NF-κB–PLAC8–BCL-xL Axis
Mechanistically, the study identified NF-κB (p65) as an upstream regulator of PLAC8.
Key findings included:
- NF-κB activation increased progressively during cadmium-induced transformation.
- Two NF-κB binding sites were identified in the PLAC8 promoter.
- Chromatin immunoprecipitation confirmed p65 binding to the PLAC8 promoter in CTPE cells.
- NF-κB knockdown reduced PLAC8 expression and restored LC3B–LAMP1 colocalization.
PLAC8, in turn, selectively upregulated the survival gene BCL-xL. Silencing PLAC8 or NF-κB reduced tumor growth in xenograft models. Inhibition of BCL-xL similarly suppressed tumor progression and reactivated autophagy- and apoptosis-related pathways.
These findings establish an oncogenic signaling axis in which chronic cadmium exposure activates NF-κB, stabilizes PLAC8, disrupts autophagy, and promotes BCL-xL–mediated survival.
Imaging as Mechanistic Evidence
A critical component of the study was the quantitative assessment of protein colocalization.
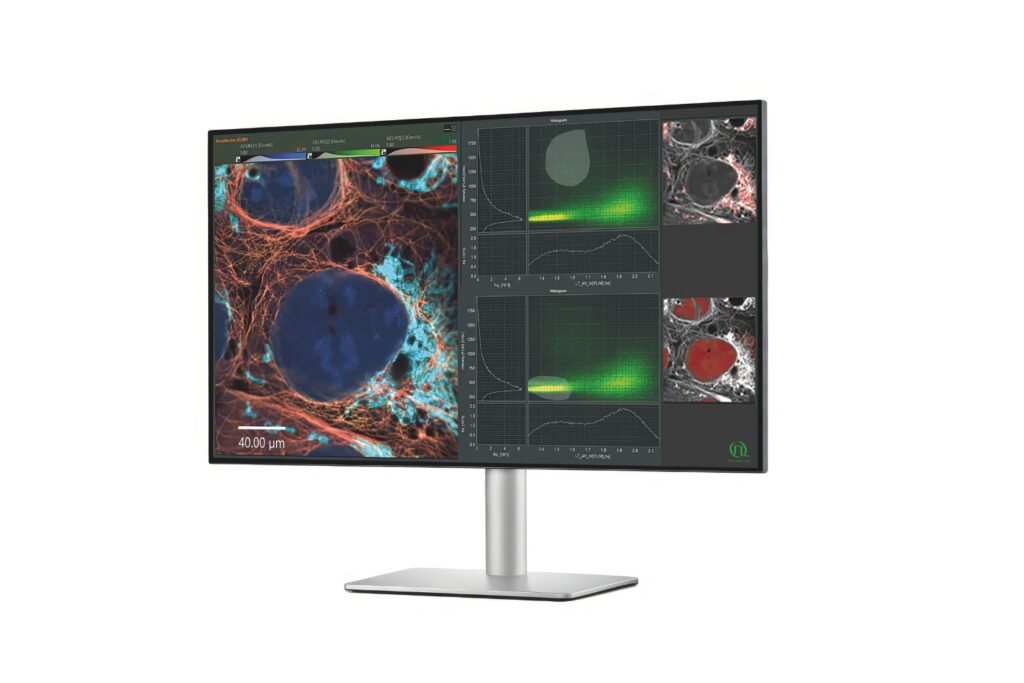
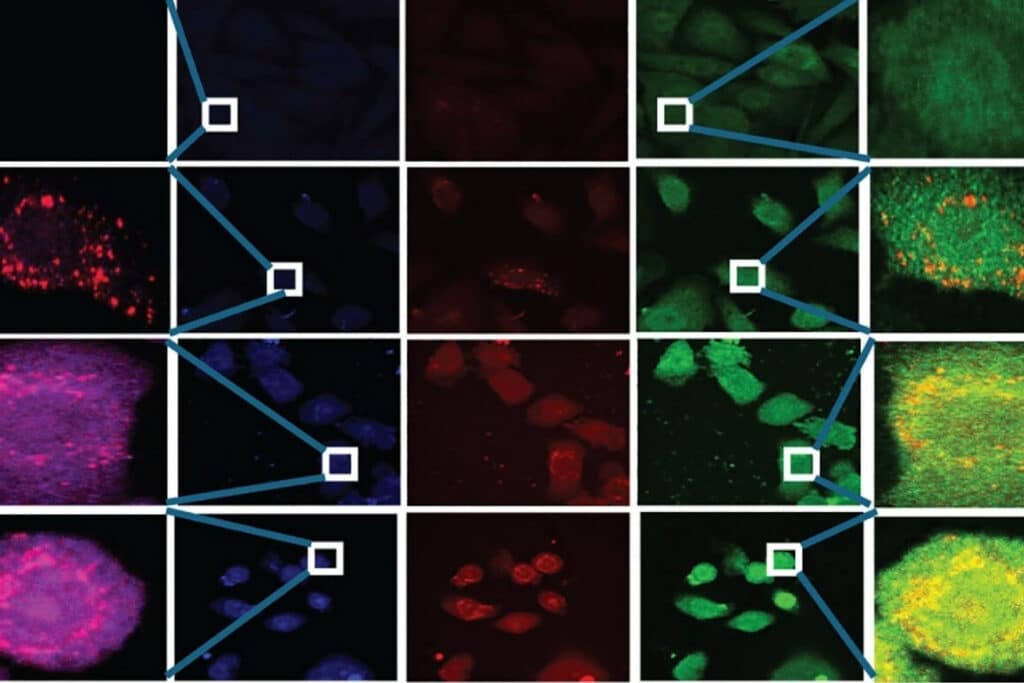
The experimental question was not simply whether LC3B, LAMP1, and PLAC8 were expressed, but whether their spatial relationships changed during transformation. This required multicolor immunofluorescence labeling combined with subcellular spatial resolution, quantitative colocalization analysis at the single-cell level, and statistical evaluation using Pearson correlation coefficients.
The study systematically compared LC3B–LAMP1 and LC3B–PLAC8 colocalization across transformation stages, demonstrating a progressive shift in fusion events. This spatial redistribution provided mechanistic evidence for defective autophagy during transformation.
Without quantitative confocal imaging, this replacement mechanism could not have been resolved with the same spatial precision.
Instrumentation Used in This Study by PicoQuant
Luminosa Single Photon Counting Confocal Microscope
In this study, the Luminosa single photon counting confocal microscope was used for multicolor fluorescence imaging and quantitative colocalization analysis. The experimental design required detection of spatially overlapping fluorescence signals within densely labeled intracellular compartments. Accurate quantification of LC3B–LAMP1 and LC3B–PLAC8 colocalization further depended on stable multichannel detection and sufficient subcellular spatial resolution. The system’s single-photon counting–based signal acquisition and multichannel fluorescence detection supported quantitative colocalization analysis using established statistical methods such as Pearson correlation. These technical characteristics enabled measurement of spatial protein redistribution during cadmium-induced transformation.

Key benefits of Luminosa:
- Reliable measurement precision
- Time-saving workflows
- Intuitive user experience
- Quick results through automated analysis
- Consistent, high-quality results
- Expert-level application support
Translational Relevance
Beyond in vitro findings, the study demonstrated:
- Reduced tumor burden following PLAC8 or NF-κB knockdown in xenograft models
- Decreased BCL-xL expression upon disruption of the signaling axis
- Increased autophagy and apoptosis pathway activation following axis inhibition
Analysis of clinical prostate cancer specimens with elevated cadmium levels showed increased expression of NF-κB, PLAC8, and BCL-xL, consistent with the experimental model. TCGA data further supported upregulation of PLAC8 and BCL-xL in metastatic prostate cancer samples with high p65 expression.
The findings suggest that the NF-κB–PLAC8–BCL-xL axis represents a mechanistically defined target for intervention in cadmium-associated prostate carcinogenesis.
Conclusion
This study provides experimental and preclinical evidence that chronic cadmium exposure drives prostate carcinogenesis through coordinated disruption of autophagy and activation of survival signaling. By quantitatively resolving protein colocalization shifts during transformation, confocal imaging was instrumental in defining the mechanistic basis of autophagy impairment. The findings demonstrate how spatially resolved fluorescence analysis can uncover regulatory interactions that are not apparent from expression measurements alone.
Learn more about the Luminosa Single Photon Counting Confocal Microscope used in this study.