Photon Counting Detectors


Complete confocal fluorescence microscope that empowers researchers to advance quantitative functional imaging from individual molecules to cells and tissues.

Modular, customizable, time-resolved confocal microscope with single-molecule sensitivity for life and materials science.

Compact FLIM and FCS upgrade kit that adds advanced functional imaging and correlation analysis to existing laser scanning microscopes.

Designed for flexible, sensitive, and precise steady-state and time-resolved spectroscopy across the UV to NIR range and time scales from picoseconds to milliseconds.

Modular lifetime spectrometer designed for flexible fluorescence and photoluminescence measurements in both materials and life science research.

Add spectral and time-resolved photoluminescence to your setup through flexible microscope–spectrometer coupling options.

Get the most out of superconducting nanowire detectors in large-scale quantum communication and computing experiments requiring precise multichannel timing.

Boost your time-resolved experiments with a flexible, high-precision time tagging and TCSPC unit for materials science and quantum sensing.

Scale your photonic quantum computing and detector characterization setups while maintaining performance, flexibility, and high data throughput.

Compact 3-color picosecond laser delivering flexible ns to ms excitation with cost-effective multicolor performance and straightforward operation.

Smart picosecond laser diode heads covering UV-A to NIR, providing the right combination of power, pulse width, and diode type for any time-resolved technique.

VisUV provides clean short pulses and stable timing across key UV and visible wavelengths, including deep UV lines as well as 488 nm and 532 nm.

Enhance your single-photon counting experiments with wide dynamic range and excellent timing precision in the UV and visible even at the highest count rates.

Capture even the weakest signals over large areas with maximum dynamic range and enhanced low-light sensitivity in a compact detector design.

Unlock spatially resolved single-photon detection with a 23-pixel SPAD array, combining low dark counts and precise time tagging for advanced experiments.
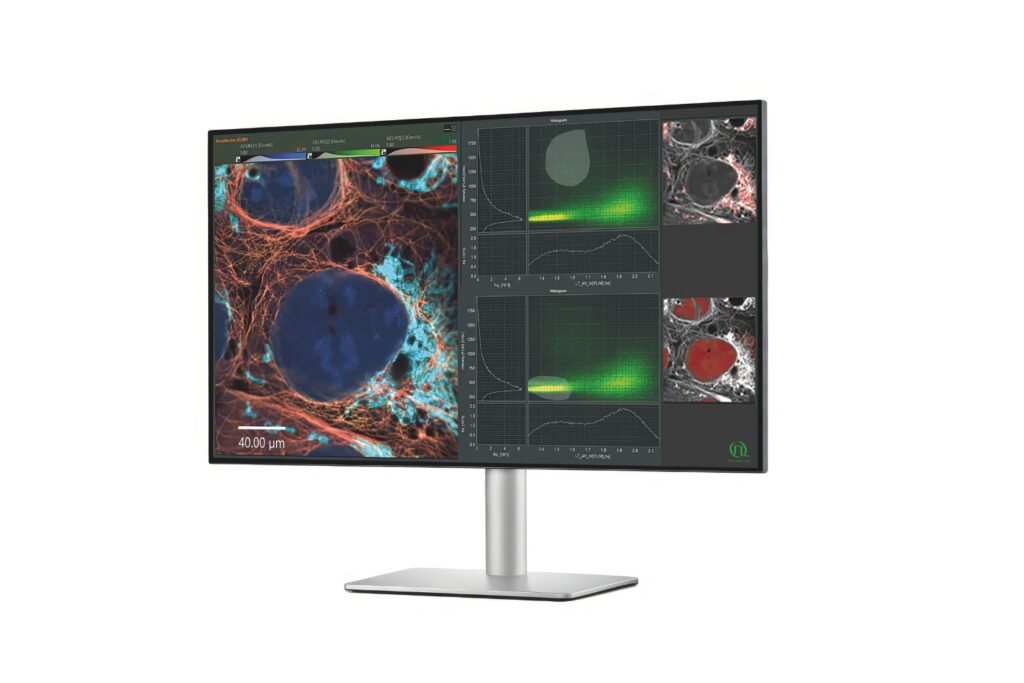
Advanced FLIM analysis software for fast, accurate interpretation of lifetime imaging data.
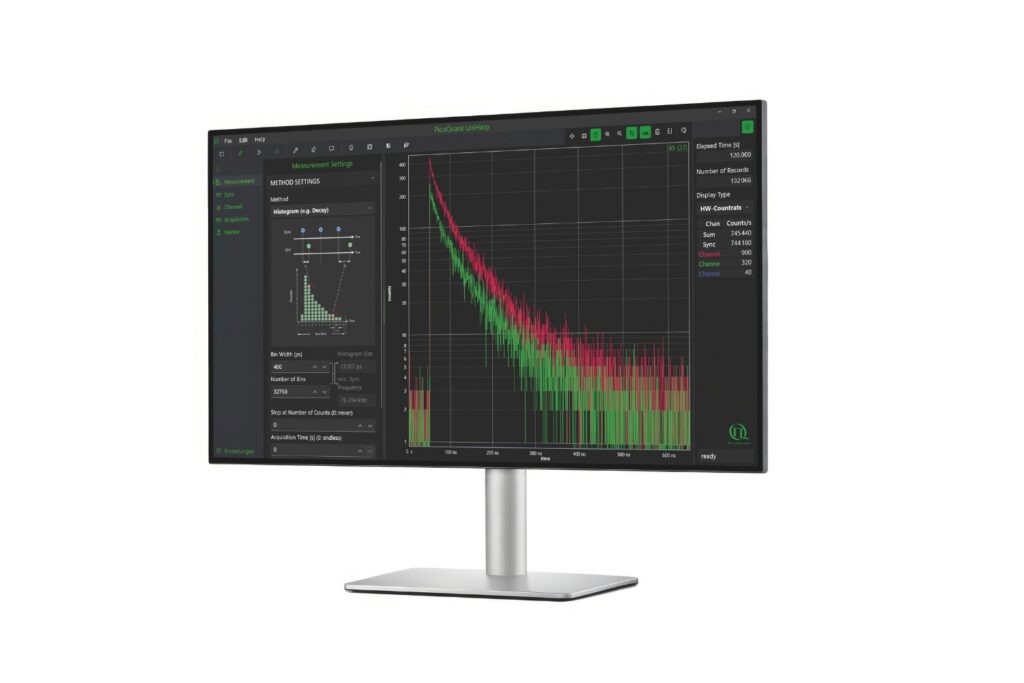
Intuitive, free software solution for real-time, high-precision photon data acquisition, visualization, and initial data analysis.
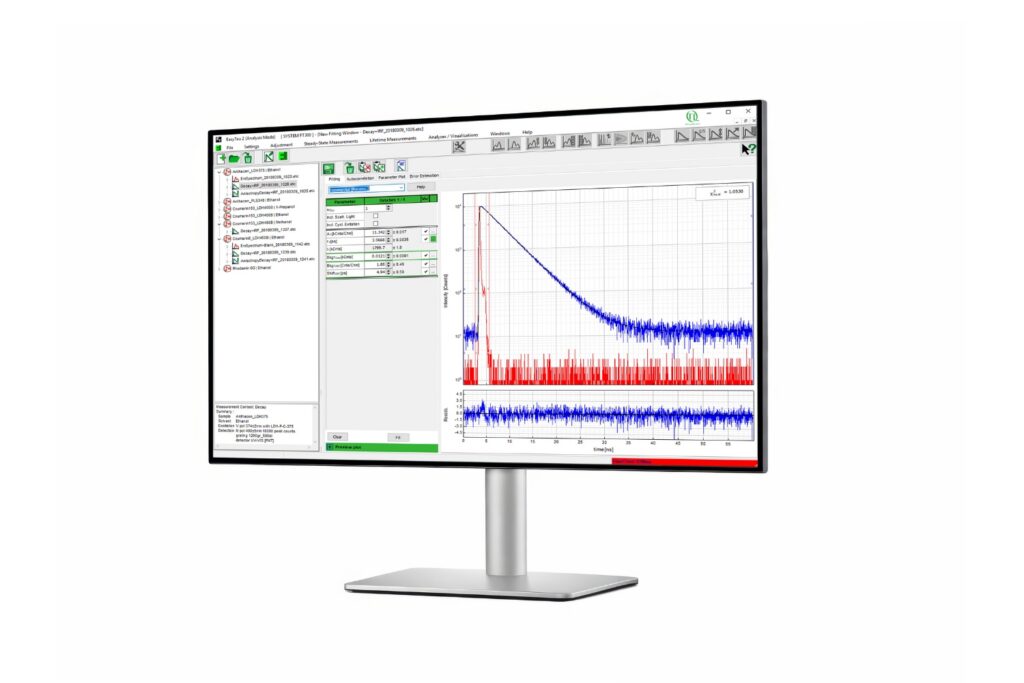
Advanced software for time-resolved fluorescence acquisition and analysis.

An imaging technique that uses fluorescence lifetimes to generate image contrast.
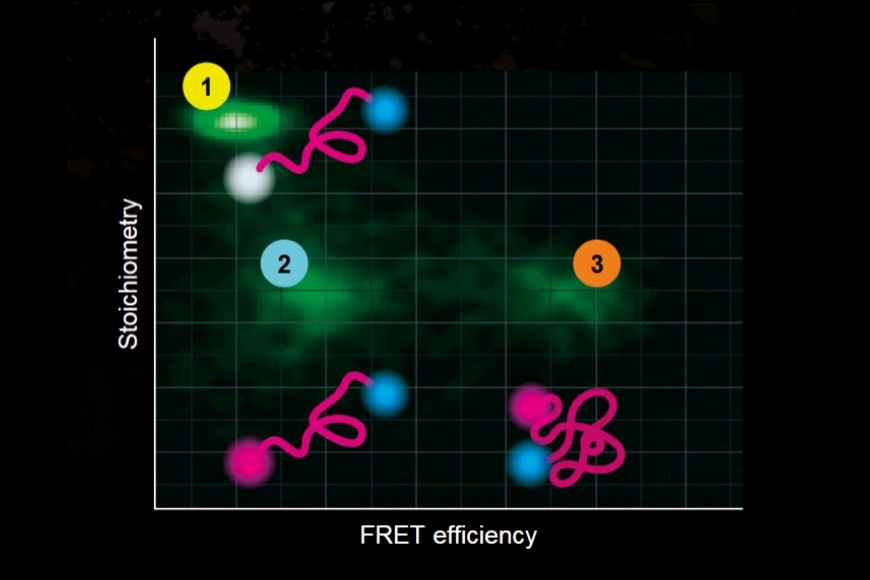
Investigating how proteins dynamically explore multiple conformational states that control biological function.

Investigating how biomolecules separate into dynamic liquid phases to organize cellular space and regulate biological function.
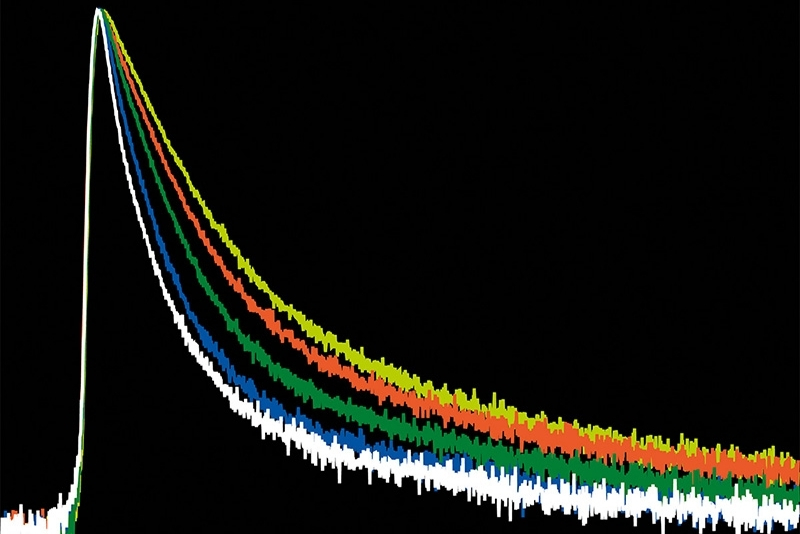
A time-resolved technique that measures photoluminescence lifetimes to reveal excited-state dynamics in materials.
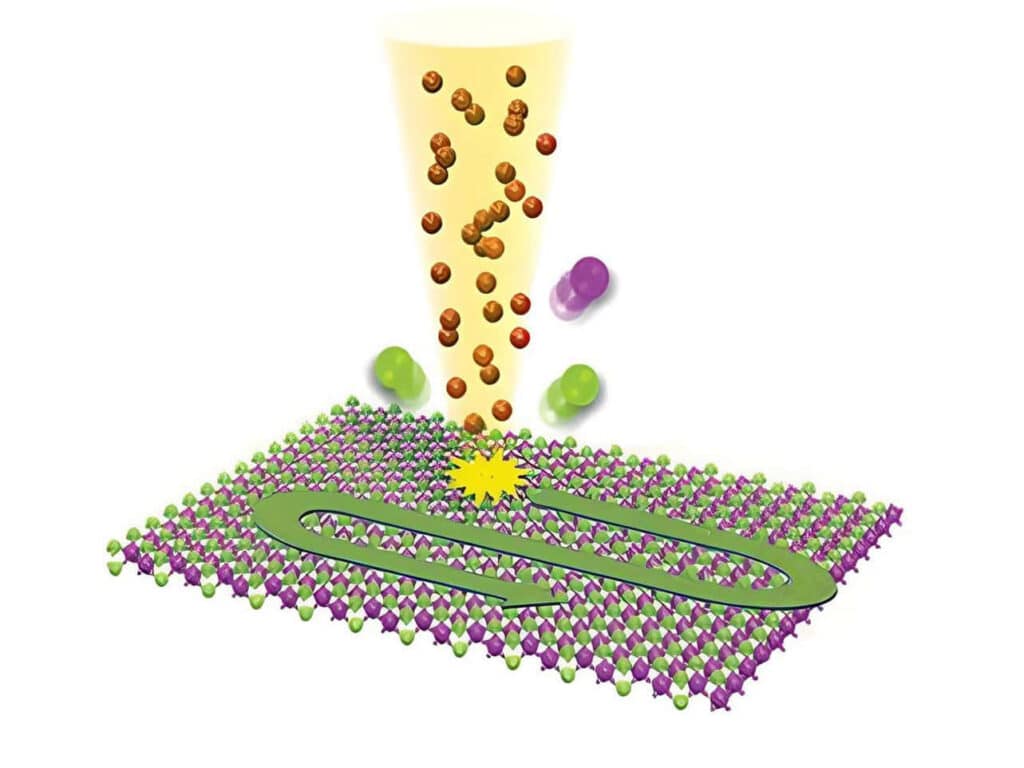
Studying exciton dynamics, charge carrier processes, and structural properties through optical and time-resolved characterization methods.

Investigating charge-carrier lifetimes and recombination dynamics to enable precise optical characterization of material quality and device performance.
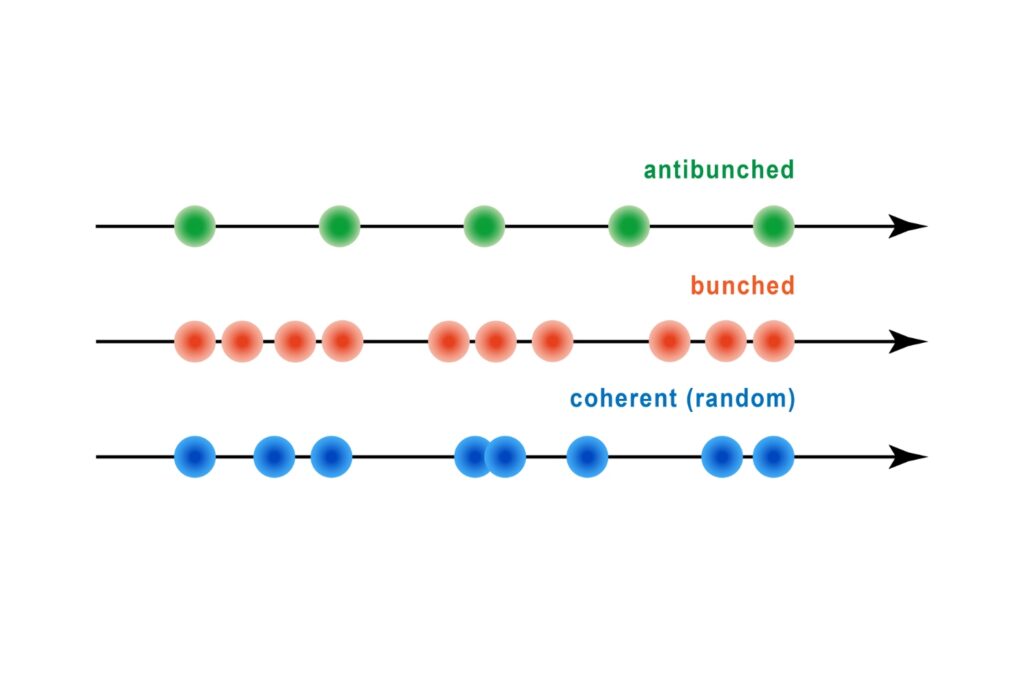
A quantum optical signature revealed by time-resolved photon correlation analysis to identify single-photon emission in materials and nanostructures.
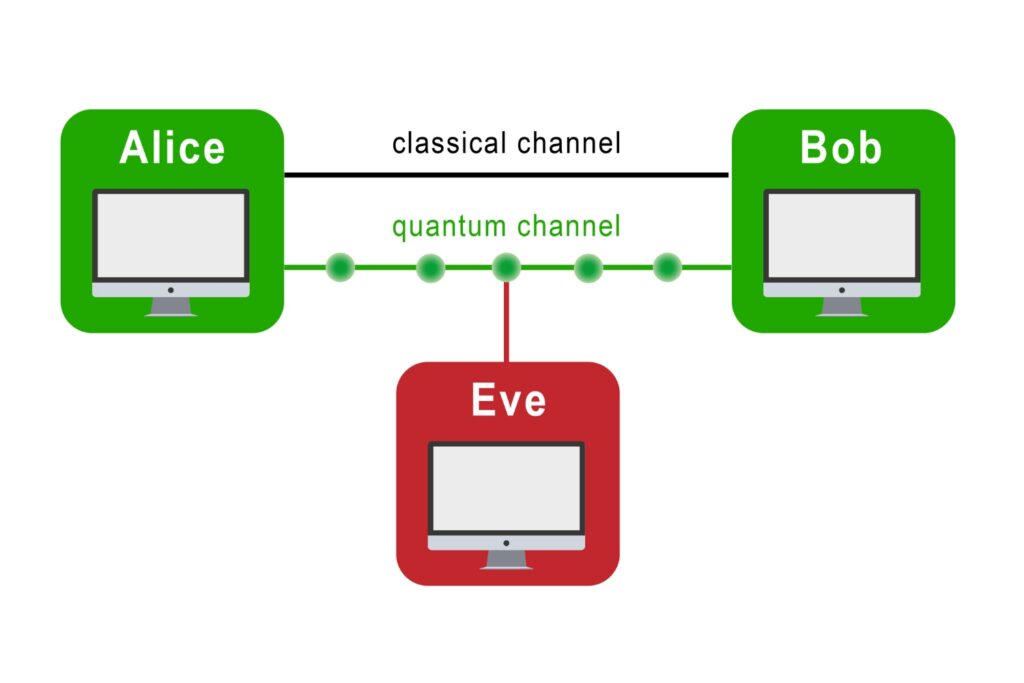
The transmission of information using individual photons, using quantum effects to ensure absolute security.
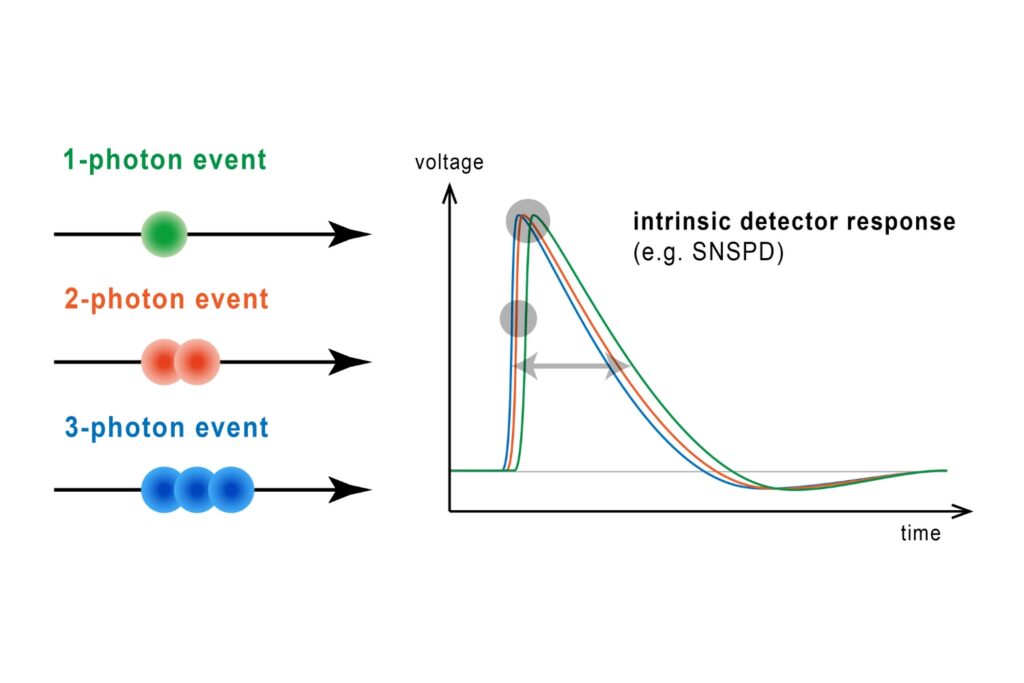
Quantifying photons per detection event enables direct access to photon-number statistics, providing insight into quantum and statistical properties of light.

An optical technique that analyzes light emission under electrical excitation to reveal electronic properties of electroluminescent materials.
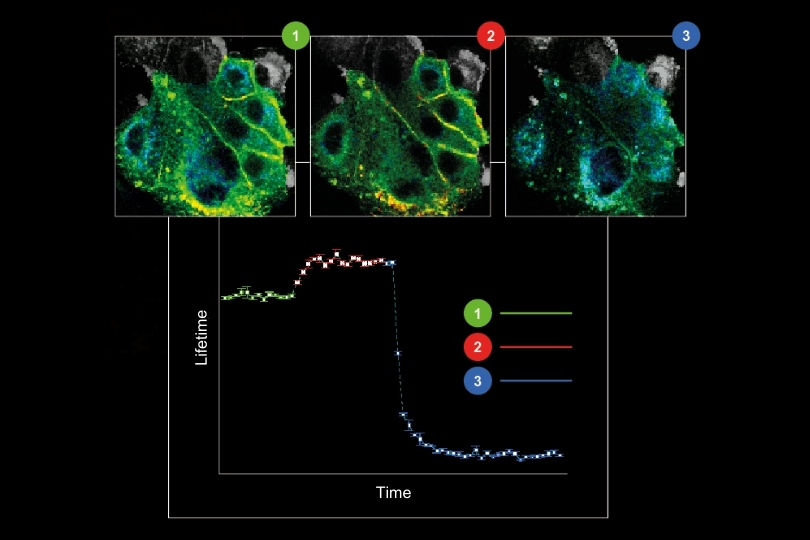
Monitoring environmental signals and trace compounds to understand dynamic changes in natural and engineered environments.
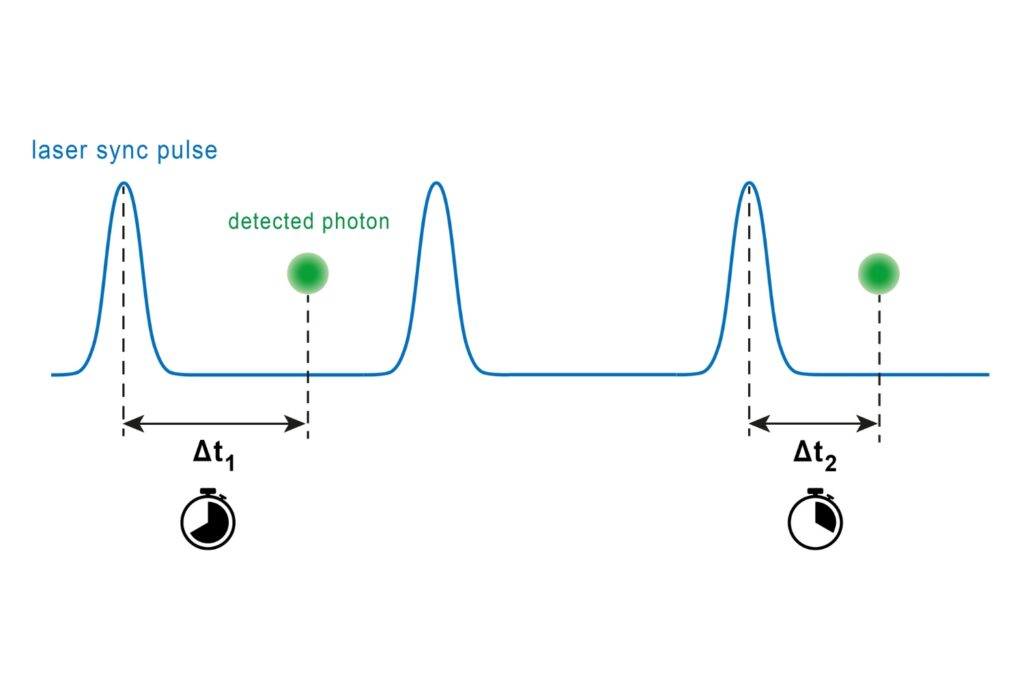
A photon timing technique that measures single-photon arrival times to resolve ultrafast dynamics in fluorescence, materials research, and quantum optics.

Nanomaterials research addresses how reducing material dimensions to the nanometer scale fundamentally alters physical behavior. Confinement, large surface-to-volume ratios, and interface effects dominate the optical and electronic properties of these systems, often overriding bulk characteristics. Research in this field spans inorganic, organic, and hybrid nanostructures and focuses on linking synthesis, morphology, and environmental factors to measurable optical signatures that reveal structure–property relationships inaccessible at larger length scales.
Optical responses of nanomaterials are governed by quantum confinement, dielectric contrast, and surface states. Absorption and emission spectra often broaden or shift due to size dispersion and local heterogeneity. Beyond steady-state observables, photophysical processes such as exciton formation, carrier trapping, and radiative or nonradiative recombination occur on fast timescales and are highly sensitive to nanoscale morphology, interfacial structure, and the surrounding environment.
After optical excitation, charge carriers in nanomaterials undergo relaxation pathways that differ from bulk systems. Nonradiative recombination, surface trapping, and energy transfer frequently dominate carrier lifetimes and energy dissipation. These processes depend on particle size, defect density, and interfacial coupling. Time-resolved measurements provide direct access to carrier lifetimes and relaxation kinetics, enabling quantitative insight into loss mechanisms that limit optical efficiency and overall material performance.
Nanomaterials are often structurally diverse. Variations in size, shape, crystallinity, and surface chemistry lead to spatially heterogeneous optical behavior. Interfaces between nanostructures and their substrates or surrounding matrices introduce additional electronic states that influence emission efficiency, charge transfer, and carrier mobility. Defects can act as quenching centers or localized emitters, making spatially resolved optical characterization essential for correlating structural variations with photophysical response.
Optical characterization techniques provide non-contact access to intrinsic nanomaterial properties. Steady-state (PL) and time-resolved photoluminescence (TRPL) reveal emission pathways, carrier lifetimes, and recombination dynamics. Imaging-based approaches extend these measurements by resolving spatial variations across individual nanostructures or ensembles. The combination of spectral, temporal, and spatial information enables a comprehensive understanding of nanoscale optical behavior under realistic experimental conditions.
Carbon dots are nanoscale carbon-based materials with size-dependent optical properties and strong, tunable photoluminescence. Their emission behavior is highly sensitive to surface chemistry, local environment, and coupling to other nanostructures, making them attractive model systems for nanomaterials research.
Monolayer MoS₂ and WSe₂ on flexible substrates were investigated using reflection imaging, SHG, TRPL, and two-photon excitation TRPL. Combining linear and nonlinear optical contrasts within a single microscope enables local correlation of structure, symmetry, and emission dynamics in two-dimensional dichalcogenide monolayers.
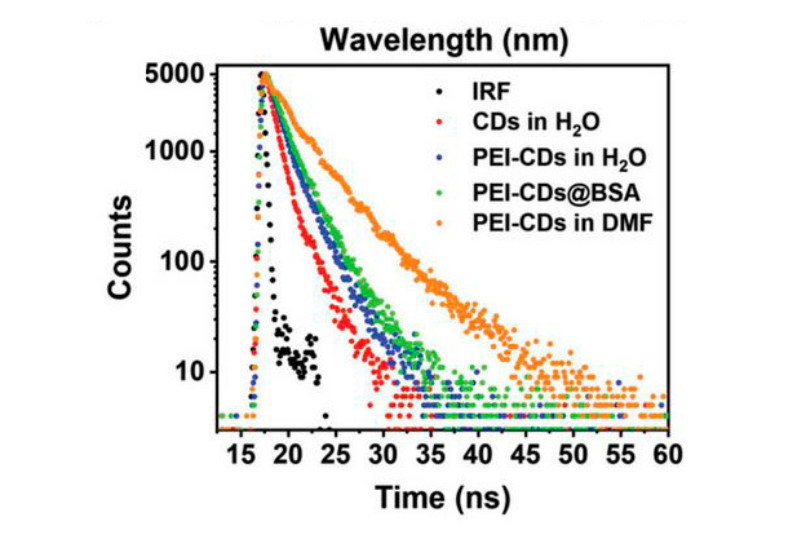
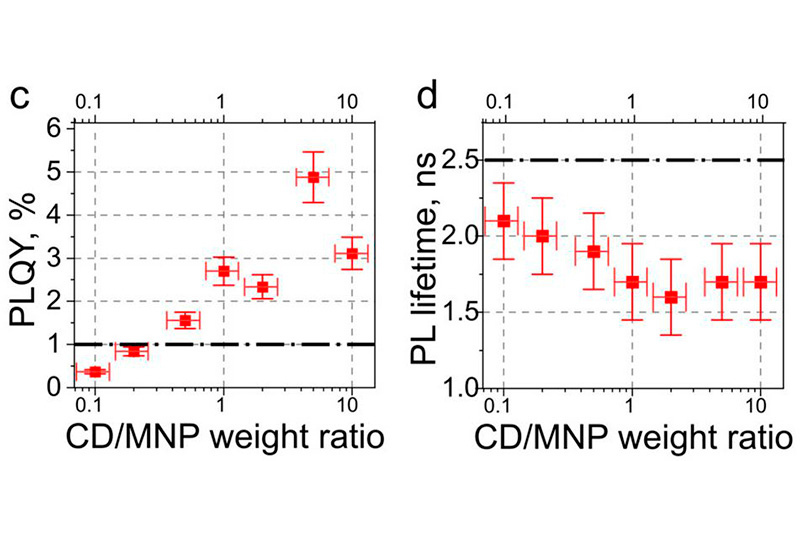
Surface modification of carbon dots enables photoluminescence in the near-infrared spectral range. Time-resolved photoluminescence measurements show increased lifetimes after polyethyleneimine functionalization, suggesting reduced nonradiative energy dissipation and improved excited-state stability in aqueous environments.
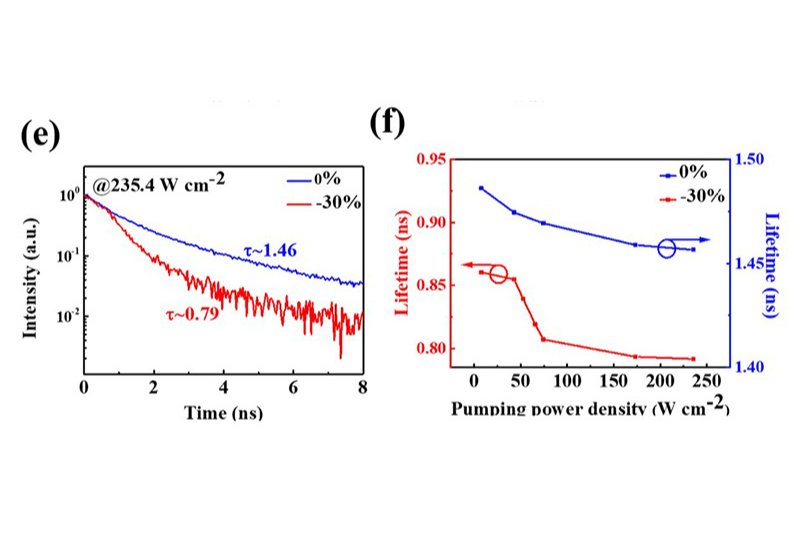
Hybrid devices combining fluorescent carbon dots with crumpled graphene enable cavity-free white laser emission. Power-dependent TRPL measurements reveal decreasing carrier lifetimes under increasing excitation, consistent with stimulated emission. Graphene-induced scattering is thought to enhance optical feedback within the nanostructured architecture.

Webinar on quantum optics in 2D TMD materials featuring multimodal imaging with MicroTime 100 and FluoTime 300 to study photoluminescence and light–matter interactions.
Second-harmonic generation imaging with picosecond lasers reveals crystal structure, defects, and layer orientation in advanced materials.
In this video, Sam Stranks (University of Cambridge), gives an overview of halide perovskite solar cell research using multimodal microscopy to study luminescence, recombination, degradation, and pathways to improve efficiency and stability.
How time-resolved fluorescence spectroscopy and microscopy reveal excited-state dynamics, defects, and charge-carrier processes
TRPL reveals energy transfer processes, lifetimes, and spatially resolved optical properties
Poster on high-spatial photoluminescence studies of nanostructures and quantum emitters using time-resolved confocal microscopy and spectroscopy.
Please fill out the form below to receive the requested file. After submitting your details, the file will be sent to you by email.
* Required
Please fill out the form below to request more information. You may also use it to inquire about pricing, availability, technical specifications, or discuss your specific application. Our sales team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information about our products and services. You may also use it to ask for pricing, availability, technical specifications, or any other details relevant to your inquiry. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required
Please fill out the form below to request more information and prices about our product. Our team will be happy to review your request and get in touch with you. If additional information is needed to process your inquiry, we will let you know.
* Required